If the heat capacity of a substance is correctly represented by an equation of the form, C
Question:
If the heat capacity of a substance is correctly represented by an equation of the form, CP = A + BT + DT−2 show that the error resulting when ⟨CP⟩H is assumed equal to CP evaluated at the arithmetic mean of the initial and final temperatures is:
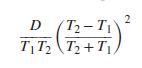
Transcribed Image Text:
D (T2- T1 TT, (T2+T|
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The error resulting when CPH is assumed equal to CP e...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
The heat capacity of anhydrous potassium hexacyanoferrate (II) varies with temperature as follows: TIK Cp,m/J K-I mol-I) TIK Cp,m/JK-I mol-I) 10 2.09 100 179.6 20 14.43 110 192.8 30 36.44 150 237.6...
-
The compressibility / of a substance is defined as the fractional change in volume of that substance for a given change in pressure:
-
The molar heat capacity of a diatomic molecule is 29.1 J/K mol. Assuming the atmosphere contains only nitrogen gas and there is no heat loss, calculate the total heat intake (in kilojoules) if the...
-
Assume your company shows the market values of equity and debt at the level of $175373 and $224626, respectively. The rate of return on assets is 33 percent and its volatility is 45 percent. The...
-
What do you think accounts for the success of Ikea (see Management Close-Up 2.2) around the world? Can other struggling U.S. retailers such as K-Mart and Sears imitate Ikeas success? Explain your...
-
Fixed costs are often defined as fixed over the short run. Does this mean that they are not fixed over the long run? Why or why not?
-
Explain precondition and post-condition by examples.
-
Members of the board of directors of Security Force have received the following operating income data for the year just ended: Members of the board are surprised that the industrial systems product...
-
Discuss what is currently happening at Cape union mart in terms of digital marketing strategy and what they should do to increase company revenue whilst maintaining service levels and building online...
-
Why do you need wireless services when adequate wired infrastructure exists in most parts of the United States?
-
If the heat capacity of a substance is correctly represented by an equation of the form, C P =A+BT+CT 2 show that the error resulting when C P H is assumed equal to C P evaluated at the arithmetic...
-
A process stream is heated as a gas from 25C to 250C at constant P. A quick estimate of the energy requirement is obtained from Eq. (4.3), with CP taken as constant and equal to its value at 25C. Is...
-
Repeat the above exercise, but this time replace the terminal value by an EBITDA (earnings before interest, taxes, depreciation, and amortization) ratio times year-5 anticipated EBITDA. Show a graph...
-
Modify BST to add a method rangeCount() that takes two keys as arguments and returns the number of keys in a BST between the two specified keys. Your method should take time proportional to the...
-
Create a new constructor for the linked-list implementation of Stack so that Stack \(t=\) new Stack (s); makes \(t\) a reference to a new and independent copy of the stack s. You should be able to...
-
Write a Digraph client TransitiveClosure whose constructor takes a Digraph as an argument and whose method isReachable (v, w) returns true if there exists some directed path from \(v\) to \(w\), and...
-
Develop a data type for a buffer in a text editor that implements the following API: public class Buffer Buffer () void insert(char c) char delete() void left(int k) void right(int k) int size()...
-
Write a recursive function that takes the first Node in a linked list as an argument and reverses the list, returning the first Node in the result.
-
(a) Show that the vectors are linearly independent. (b) Show that they also span R4. (c) Write (1.0. 0, I )T as a linear combination of them. 9008
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
Natural gases arc rarely pure methane; they usually also contain other light hydrocarbons and nitrogen. Determine on expression for the standard heat of combustion as a function of composition for a...
-
If the heat of combustion of urea, (NH2)2CO(s), at 25(C is 631 660 J mol-1 when the products are CO2(g), H2O(l), and N2(g), what is (H((298 for urea?
-
A light fuel oil with an average chemical composition of C10H18 is burned with oxygen in a bomb calorimeter. The heat evolved is measured as 43,960 J g-1 for the reaction at 25 (C. Calculate the...
-
For each ordered pair, determine -6x+7y=2 7x-4y=6 a solution to the system of equations.
-
Review the meaning of the concepts or terms given in Key Terms and Concepts. Distinguish between a production department and a service department. Distinguish between a direct cost and an indirect...
-
What would you like the members of your learning community to know about your global health interests, perspectives, and life experiences? How will you prepare to listen and learn from others? Why...

Study smarter with the SolutionInn App


