One mole of an ideal gas with constant heat capacities undergoes an arbitrary mechanically reversible process. Show
Question:
One mole of an ideal gas with constant heat capacities undergoes an arbitrary mechanically reversible process. Show that:
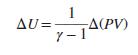
Transcribed Image Text:
AU=- 7-14(PV)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
To begin we can use the first law of thermodynamics which states that the change in internal energy ...View the full answer

Answered By

Morris Maina
I am a professional teaching in different Colleges and university to solved the Assignments and Project . I am Working more then 3 year Online Teaching in Zoom Meet etc. I will provide you the best answer of your Assignments and Project.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas with constant heat capacities undergoes a change of state from conditions T1, P1 to conditions T2, P2. Determine (H (J mol-1) and (S (J mol-1 K-1) for one of the following cases. (a) T1...
-
One mole of an ideal gas with heat capacity Cv goes through a process in which its entropy S depends on T as S = a/T, where a is a constant. The gas temperature varies from T1 to T2. Find: (a) The...
-
One mole of an ideal gas with the adiabatic exponent y goes through a polytropic process as a result of which the absolute temperature of the gas increases T-fold. The polytropic constant equals n....
-
Choose a company or product that you have been noticing or admiring. Develop a plan to advertise this product that involves the United States and at least one other country, and three forms of media....
-
Is it possible for a small business owner to take an active role in social responsibility? What are some ways that a small business (for example, a family business or a local business you are...
-
Describe two or more factors that the auditor should consider in assessing the inherent risk for (a) Intangible assets and (b) The property management process.
-
Use the Newsvendor Model spreadsheet to set up and run a Monte Carlo simulation assuming that demand is Poisson with a mean of 45 but a minimum value of 40 (use the lower cutoff parameter in the...
-
Diagrams of two self-service gasoline stations, both located on corners, are shown in Figure (a) and (b). Both have two rows of four pumps and a booth at which an attendant receives payment for the...
-
How does diversification affect systematic and unsystematic risk?
-
The Tinker Construction Company is ready to begin a project that must be completed in 12 months. This project has four activities (A, B, C, D) with the project network shown next. The project...
-
A substance for which is a constant undergoes an isothermal, mechanically reversible process from initial state (P 1 , V 1 ) to final state (P 2 , V 2 ), where V is molar volume. (a) Starting with...
-
Estimate: (a) The mass of ethane contained in a 0.15 m 3 vessel at 60C and 14,000 kPa. (b) The temperature at which 40 kg of ethane stored in a 0.15 m 3 vessel exerts a pressure 20,000 kPa.
-
Fill in the blanks with an appropriate word, phrase, or symbol(s). The sum of the measures of the interior angles of a triangle is _______.
-
Compared to other majornations, the United States spends________ on health care and achieves________ efficiency. A. more; about the same B. about thesame; less C. more; less D. less; less E. less;...
-
Studying other cultures through a humanistic lens allows people to understand how different cultures came about and how and why people behave differently from one place to another (Lombrozo, 2015)....
-
4. Assume that G and T are exogenous, and C is determined by the standard. consumption function, but that investment is now endogenous and responds to income: I = b + bY. Assume c + b < 1. (a)...
-
4. You have decided it's time to buy a house, and you have found the one you want. The price is $500,000, and you will pay 10% in cash and will take a mortgage on the balance. The annual interest...
-
Differentiate. G(x) = (2x+3) (9x+ (x) G'(x)=
-
Benjamin Company has rented new equipment to Murrell Builders that cost Benjamin $48,000, and has a fair value of $50,000. This nonspecialized equipment has a life of 4 years and no residual value at...
-
If someone's Z-score for a variable was 0.67. Their score is a significant extreme score. Their score is not significant. O Their score is slightly above average. O Their score is an outlier.
-
Determine a good estimate for the compressibility factor Z of saturated hydrogen vapor at 25 K and 3-213 bar. For companion, an experimental value is Z = 0.7757.
-
Five kilograms of liquid carbon tetrachloride undergo a mechanically reversible, iso- haric change of state at I bar during which the temperature changes from 0(C to 20(C. Determine (V1, W, , (H1,...
-
Five kilograms of liquid carbon tetrachloride undergo a mechanically reversible, iso- haric change of state at I bar during which the temperature changes from 0(C to 20(C. Determine (V1, W, , (H1,...
-
Columbus Industries makes a product that sells for $37 a unit. The product has a $29 per unit variable cost and total fixed costs of $10,000. At budgeted sales of 1,950 units, the margin of safety...
-
18. Suppose that Maxima shares are selling for $10 per share and you own a call option to buy Maxima shares at $7.50. The intrinsic value of your option is:
-
ABC Insurance Company reported the following information on its accounting statements last year: What was ABC 's expense ratio last year

Study smarter with the SolutionInn App


