The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 62 kPa.
Question:
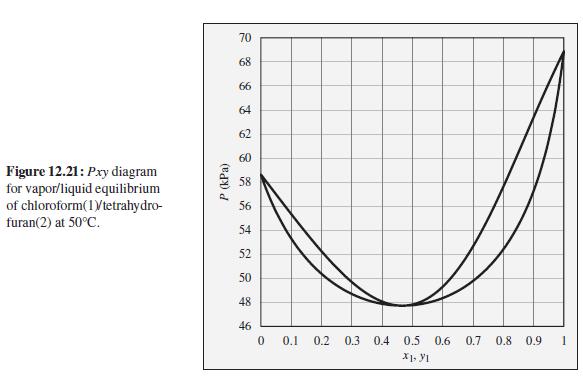
The pressure above a mixture of chloroform and tetrahydrofuran at 50°C is measured to be 62 kPa. What are the possible compositions of the liquid and vapor phases?
To the Pxy diagram for chloroform(1)/tetrahydrofuran(2) at 50°C shown in Fig. 12.21.
Transcribed Image Text:
70 68 66 64 62 60 Figure 12.21: Pxy diagram for vapor/liquid equilibrium of chloroform(1Vtetrahydro- furan(2) at 50°C. 58 56 54 52 50 48 46 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 P (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
A relativistic rocket is measured to be 50 m long, 2.5 m high, and 2.0 m wide by its pilot. It is traveling at 0.65c (in the direction parallel to its length) relative to an inertial observer. (a)...
-
A mixture of chloroform and tetrahydrofuran is heated in a closed system at 120 kPa to a temperature of 75C, and two phases are observed to be present. What are the possible compositions of the...
-
The pressure of an automobile tire is measured to be 190 kPa (gage) before a trip and 215 kPa (gage) after the trip at a location where the atmospheric pressure is 95 kPa. If the temperature of air...
-
A teacher traces a small circle on the palm of a kindergartener's hand to let him know it is time for free play. What type of disability does this child most likely have? A. Traumatic Brain Injury B....
-
1. Explain when a country has a comparative advantage over another country. 2. Use the law of comparative advantage to determine what goods countries will specialize in. 3. Explain how two countries...
-
Assuming monetary benefits of an information system at $85,000 per year, one-time costs of $75,000, recurring costs of $35,000 per year, a discount rate of 12 percent, and a five-year time horizon,...
-
You currently owe $\$ 18,000$ on a car loan at $9.5 \%$ interest. If you make monthly payments of $\$ 576.59$ per month, how long will it take you to fully repay the loan?
-
Mark each of the following statements as true or false and explain why the false statements are incorrect. a. Results benchmarking relies only on comparisons to firms within the same industry. b. SPC...
-
Headland Hardware's payroll for November is summarized below. Amount Subject to Payroll Taxes Unemployment Tax Payroll Wages Due FICA Federal State Factory $124,000 $124,000 $39,000 $39,000 Sales...
-
Given a heap H and a key k, give an algorithm to compute all the entries in H having a key less than or equal to k. For example, given the heap of Figure 9.12a and query k =7, the algorithmshould...
-
The pressure above a mixture of chloroform and tetrahydrofuran at 50C is measured to be 52 kPa. What are the possible compositions of the liquid and vapor phases? To the Pxy diagram for...
-
Consider a closed vessel of fixed volume containing equal masses of water, ethanol, and toluene at 70C. Three phases (two liquid and one vapor) are present. (a) How many variables, in addition to the...
-
Which of the following education-related expenses can be used to claim an education credit? Tuition Fees Room and Board Meals Yes Yes a. Yes Yes No b. Yes Yes Yes Yes Yes C. Yes No Yes Yes d. No No
-
In the Statement of Revenues, Expenditures, and Changes in Fund Balances, transfers must be reported a. in a separate section immediately following revenues. b. in a section immediately following the...
-
The following GAAP requirements for budgetary reporting are true except a. budgetary comparisons for the General Fund and major Special Revenue Funds must include expenditure data that is at least as...
-
Which of the following statements would be true concerning budgetary integration? a. The integration of budgetary accounts into the general ledger does not affect the asset and liability accounts. b....
-
The budget data presented in a school district General Fund statement of revenues, expenditures, and changes in fund balancesbudget and actualare to be a. the original, legally adopted budget. b. the...
-
Which of the following items does a government with a modified accrual and encumbrances basis budget report differently in its budgetary basis statement of revenues, expenditures, and changes in fund...
-
Prove that if A is a regular 2 Ã 2 matrix, then its L U factorization is unique. In other words, if where L, are special lower triangular and are upper triangular, then and A=LU = LU where L,L
-
For the vector whose polar components are (Vr = 1, Vθ = 0), compute in polars all components of the second covariant derivative Vα;μ;ν. To find...
-
The energy balance can be developed for just about any process. Since our goal is to learn how to develop model equations as well as to simply apply them, it is valuable practice to obtain the...
-
Steam undergoes a state change from 450C and 3.5 MPa to 150C and 0.3 MPa. Determine H and U using the following: (a) Steam table data. (b) Ideal gas assumptions. (Be sure to use the ideal gas heat...
-
The energy balance can be developed for just about any process. Since our goal is to learn how to develop model equations as well as to simply apply them, it is valuable practice to obtain the...
-
The controller for Tulsa Medical Supply Company has established the following activity cost pools and cost drivers. Machine setups Budgeted Overhead Cost Cost Driver Number of setups Weight of raw...
-
In 2023, Miranda records net earnings from self-employment of $168,500. She has no other income. Determine the amount of Miranda's self-employment tax and her AGI income tax deduction. In your...
-
Describe how empowerment, work groups, and multifunctional teams would or would not affect the five types of problems.

Study smarter with the SolutionInn App


