The pressure above a mixture of ethanol and ethyl acetate at 70C is measured to be 86
Question:
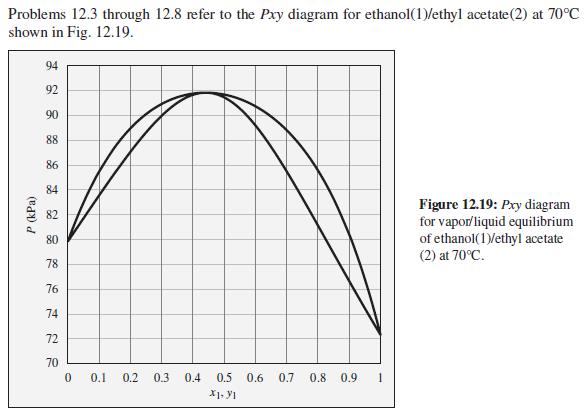
The pressure above a mixture of ethanol and ethyl acetate at 70°C is measured to be 86 kPa. What are the possible compositions of the liquid and vapor phases?
Transcribed Image Text:
Problems 12.3 through 12.8 refer to the Pxy diagram for ethanol(1)/ethyl acetate(2) at 70°C shown in Fig. 12.19. 94 92 90 88 86 84 Figure 12.19: Pxy diagram for vapor/liquid equilibrium of ethanol(1)/ethyl acetate (2) at 70°C. 82 80 78 76 74 72 70 0.1 0.2 0.3 0.4 0.5 0.6 0.7 0.8 0.9 1 P (kPa)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
To determine the possible compositions of the liquid and vapor phases we need to use a phase equilibrium calculation based on the given pressure and t...View the full answer

Answered By

BillClinton Muguai
I have been a tutor for the past 5 years. I have experience working with students in a variety of subject areas, including computer science, math, science, English, and history. I have also worked with students of all ages, from elementary school to college. In addition to my tutoring experience, I have a degree in education from a top university. This has given me a strong foundation in child development and learning theories, which I use to inform my tutoring practices.
I am patient and adaptable, and I work to create a positive and supportive learning environment for my students. I believe that all students have the ability to succeed, and it is my job to help them find and develop their strengths. I am confident in my ability to tutor students and help them achieve their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
A relativistic rocket is measured to be 50 m long, 2.5 m high, and 2.0 m wide by its pilot. It is traveling at 0.65c (in the direction parallel to its length) relative to an inertial observer. (a)...
-
A mixture of ethanol and 1-propanol behaves ideally at 36C and is in equilibrium with its vapor. If the mole fraction of ethanol in the solution is 0.62, calculate its mole fraction in the vapor...
-
The pressure of an automobile tire is measured to be 190 kPa (gage) before a trip and 215 kPa (gage) after the trip at a location where the atmospheric pressure is 95 kPa. If the temperature of air...
-
1. Suppose that a stoichiometric mixture of isooctane (C8H18) and air is burned in an engine and then the fuel is changed to 10% (by liquid volume) ethanol and 90% by liquid volume isooctane. If the...
-
The lowest fifth of income earners have a 10 percent income share; the second fifth, a 17 percent income share; the third fifth, a 22 percent income share; the fourth fifth, a 24 percent income...
-
In classical plate theory, for a circular plate with radius a and thickness A, the governing equation can be written as d 1 2 - (+) (+)- dra r dr (1) where w is the vertical deflection, P is the...
-
The price of a share is \(\$ 40\), and it is incremented in \(6 \%\) or it goes down in \(5 \%\) every three months. If the risk-free interest rate is \(8 \%\) per year, continuously compounded,...
-
A companys simplified balance sheet and income statement follow. Total assets and owners equity at the beginning of 2014 were $180,000 and $140,000, respectively. The owner made no investments or...
-
es AA 4-3 Extended Analysis LO A1 The following selected information is available from Samsung's financial statements. In millions Current assets Current liabilities Required: Current Year Prior Year...
-
What is the citation of a 1993 ALR annotation that discusses the admissibility of polygraph test results in an action for malicious prosecution? What Am. Jur. 2d evidence research references are...
-
Of the following binary liquid/vapor systems, which can be approximately modeled by Raoults law? For those that cannot, why not? Table B.1 (App. B) may be useful. (a) Benzene/toluene at 1(atm). (b)...
-
A stream of 12 kgs 1 of Cu(NO 3 ) 2 6H 2 O and a stream of 15 kgs 1 of water, both at 25C, are fed to a tank where mixing takes place. The resulting solution passes through a heat exchanger that...
-
How does contingency management help to explain why some companies succeed during tough economic environments while others fail?
-
A parent and a subsidiary continue to exist as separate legal entities even though they are economically one reporting entity. Is this true or false?
-
Article-based Discussion Read the article Z Scores A Guide to Failure Prediction, by G.J. Eidleman, The CPA Journal, February 1995. Required: 1. What judgments are involved in the Z-score? 2. What...
-
To test whether a bonus plan will improve the monthly sale volume in units, the monthly sale volumes of six salespersons before and after a bonus plan were recorded. At 99 % confidence, determine...
-
A production manager is interested in the number of defects in batches derived from different production processes. He examines a random sample drawn from each process and records the following data:...
-
Refer to question 41 to find the power of a 10 % level test when the true population mean mileage is 36 miles per gallon. Question 41 An automobile manufacturer claims that a new car gets an average...
-
Prove that an n n matrix with complex entries is unitary if and only if its rows form an orthonormal set in Cn with the Euclidean inner product.
-
Use this circle graph to answer following Exercises. 1. What fraction of areas maintained by the National Park Service are designated as National Recreation Areas? 2. What fraction of areas...
-
Data from the Bureau of Standards (J. Phys. Chem. Ref. Data, vol. 11, suppl. 2, 1982) include the following heats of formation for 1 mol of C a Cl 2 in water at 25C: From these data prepare a plot of...
-
The standard heat of reaction H for gas-phase reactions is independent of the choice of standard-state pressure P. (Why?) However, the numerical value of G for such reactions does depend on P. Two...
-
Equilibrium at 425 K and 15 bar is established for the gas-phase isomerization reaction n-C 4 H 10 (g) iso-C 4 10 (g) Estimate the composition of the equilibrium mixture by two procedures: (a)...
-
A 7 5 kg cliff - diver is falls from a height through the air head, diving towards the water. The drag coefficient for the diver is 0 . 8 3 and the area of the descending diver is 0 . 2 1 m ^ 2 ....
-
In the automobile industry, the dimensionless drag coefficient and the area of the vehicle are often combined into one variable - the drag area whereby the drag area is the product of the...
-
When you drop 10 pebbles into the well, you record the times for hearing the splash as 2.94 s, 3.11 s, 3.12 s, 2.97 s, 3.12 s, 2.97 s, 3.06 s, 3.21 s, 3.37 s, and 3.53 s. (a) Find the average time...

Study smarter with the SolutionInn App


