Determine the model parameters for the two-suffi x and the three-suffi x Margules equations using the liquidvapor
Question:
Determine the model parameters for the two-suffi x and the three-suffi x Margules equations using the liquid–vapor equilibrium data for a binary system of benzene (1)–cyclohexane (2) at 10°C. Mole fractions of liquid vs. total pressure are reported in Table E8.9A.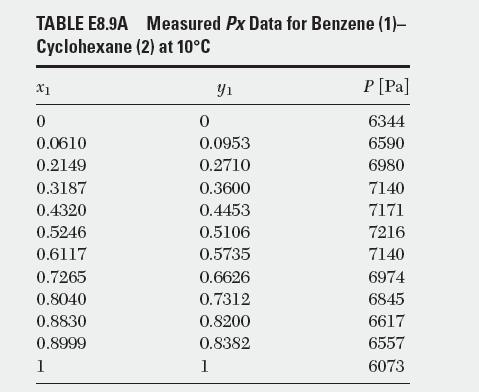
Transcribed Image Text:
TABLE E8.9A Cyclohexane X1 0 0.0610 0.2149 0.3187 0.4320 0.5246 0.6117 0.7265 0.8040 0.8830 0.8999 1 Measured Px Data for Benzene (1)- (2) at 10C Y 0 0.0953 0.2710 0.3600 0.4453 0.5106 0.5735 0.6626 0.7312 0.8200 0.8382 1 P[Pa] 6344 6590 6980 7140 7171 7216 7140 6974 6845 6617 6557 6073
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The excess Gibbs energy can be written g RTx ln y x ln y RT x In 1 RT 2 la 5 x la 5 In XPat where we ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Calculate the three-suffi x Margules parameters, A and B, for the system of Example 8.9. Example 8.9 Liquidvapor equilibrium data have been collected for a binary system of benzene (1)cyclohexane (2)...
-
Liquidvapor equilibrium data have been collected for a binary system of benzene (1)cyclohexane (2) at 10C. Mole fraction of liquid and vapor vs. total pressure are reported in Table E8.9A.5 From...
-
A binary system of species 1 and 2 consists of vapor and liquid phases in equilibrium at temperature T for which In y1 = 1.8x22 In y2 = 1.821. P1sat = 1.24 bar P2sat = 0.89 bar. Assuming the...
-
How do standard costs illustrate the trade-off between decision making and control?
-
When preparing a statement of cash flows wider the indirect method, an increase in ending accounts receivable over beginning accounts receivable will result in an adjustment to net income in the...
-
Consider f(x) = ln (1 - x) and its Taylor polynomials given in Example 8. a. Graph y =|f(x) - p 2 (x)| and y = |f(x) - p 3 (x)| on the interval [-1/2, 1/2] (two curves). b. At what points of [-1/2,...
-
The ages (in years) of the residents of a small town in 2012 Approximate the mean of the frequency distribution. Age (in years) Frequency 0-9 40 10-19 72 20-29 78 30-39 90 40-49 84 50-59 42 60-69 31...
-
Dirk Company reported the following balances at December 31, 2016: common stock $500,000, paid-in capital in excess of par value common stock $100,000, and retained earnings $250,000. During 2017,...
-
Positioning when it comes to brand and product refers to the way and space a product "takes up" in a consumer's mind as it relates to the market, their life, and competitive products (Kerin &...
-
Calculate the solubility of O2 from air in the atmosphere in equilibrium with liquid H2O at 25C. Report the answer in mole fraction and in molarity.
-
Show that the composition of the vapor and liquid phases must be equal at an azeotrope.
-
Perform the indicated operations, expressing all answers in the form a + bj. 1-j 3j
-
In theory, with all General Staff positions filled and using the maximum span of control, how many incident personnel would it be possible to manage?
-
If the farmer expresses his trade-off between cash and additional assets at the end of the planning period as $2 of cash being worth as much as $3 of additional assets, how would you incorporate this...
-
Dexter Industries purchased packaging equipment on January 8 for $290,400. The equipment was expected to have a useful life of four years, or 7,200 operating hours, and a residual value of $24,000....
-
What is missing from this positioning statement of AcceleRev? "For hospitals, AcceleRev is a revenue and accounts receivables management company that helps them speed up insurance company...
-
What would the proposed sales tax percentage paid at the register for NEW Goods and SERVICES?
-
Why are visual aids particularly useful to inexperienced speakers?
-
What are the two components of a company's income tax provision? What does each component represent about a company's income tax provision?
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b) CH CH3 : CH-C-CH-CH CH-C=N:
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
What is the Value of Employee Benefits? Discuss the Key Issues in Benefit Planning, Designing, and Administration.
-
How is the double-entry accounting system used to record financial transactions, and what are the basic principles behind it?
-
One more year passes, and your remaining $1.03 million (net of fees) is now valued at $1.10 million. What do you pay to the manager at the end of the second year?

Study smarter with the SolutionInn App


