Hydrogen cyanide can be manufactured by reaction of acetylene and nitrogen: Calculate the equilibrium composition at 800
Question:
Hydrogen cyanide can be manufactured by reaction of acetylene and nitrogen: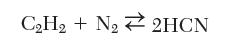
Calculate the equilibrium composition at 800 K and 1 bar.
Transcribed Image Text:
CH + N 2HCN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The following selected transactions were completed during the first year of operations: Jan. 3 Issued 15,000 shares of common stock at $23 per share for cash. 31 Issued 200 shares of common stock to...
-
Acetylene (C2H2) and nitrogen (N2) both contain a triple bond, but they differ greatly in their chemical properties. (a) Write the Lewis structures for the two substances. (b) By referring to...
-
Hydrogen cyanide, HCN, can be made by a two-step process. First, ammonia is reacted with O2 to give nitric oxide, NO. 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(g) Then nitric oxide is reacted with methane,...
-
Discuss the ways that managed care organizations can infl uence the adoption of new technologies.
-
Which deal with advocacy and lobbying; governing and managing international and global organizations; and social entrepreneurship within the non-profit organization. Think of a non-profit...
-
Use spherical coordinates. where E is the solid hemisphere x 2 + y 2 + z 2 < 9, y > 0.
-
Number of classes: 6 Data set: January production (in units) for 21 manufacturing plants of a multinational company Construct a frequency distribution and a frequency histogram for the data set using...
-
Quinn Corporation is subject to tax in States G, H, and I. Quinns compensation expense includes the following. Officers' salaries are included in the payroll factor for G and I, but not for H....
-
(1 point) A cryptography student has accidentally used the same keystream to encrypt two different messages, M and M2. We know that the ciphertexts are C 0x06c6d00e4633400a and C2 =...
-
Consider the equilibrium between copper and its oxide: The Gibbs energy of formation of Cu2O is given by Make a plot of pO2 vs. T, illustrating where Cu is stable and where Cu2O is stable in the...
-
Solve the multiple chemical reaction equilibrium problem in Example 9.19 at 800 K using the following set of independent reactions: Example 9.19 Consider a system initially charged with 1 mole of...
-
Does the black grease beneath football players? eyes really reduce glare or does it just make them look intimidating? In a variation of a study actually conducted at Yale University, 46 participants...
-
What has prompted the Texas Legislature to begin limiting the powers of home rule cities like Austin?
-
Suppose X and Y are binary random variables, each taking the values 0 and 1. If we know that the event X = x is independent of the event Y= y, i.e x 1 y, does it mean that XLY? Give mathematical...
-
Jorge wants to start a CrossFit gym. He loves to work out, has rented a metal commercial building, and has rented a metal commercial building and has purchased the necessary equipment. He will have...
-
The auditor feels that the allowance for doubtful debts should be higher than what management has estimated. In contrast to the profit before taxes, the allowance amount difference is not...
-
Harold discovers a material misstatement while auditing a client's accounts payable. The manger on the job tells Harold to ignore the error so that the client does not get upset. Harold wants to be...
-
If you are asked an illegal interview question, why is it important to first assess the intentions of the interviewer?
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
(a) Distinguish between a step and a terrace. (b) Describe how steps and terraces can be formed by dislocations.
-
Distinguish between the following adsorption isotherms: Langmuir, BET, Temkin, and Freundlich.
-
Describe the essential features of the Langmuir-Hinshelwood, Eley-Rideal, and Mars van Krevelen mechanisms for surface-catalyzed reactions.
-
Refer to the original data. The sales manager is convinced that a 1 0 % ?reduction in the selling price, combined with an increase of $ 3 5 , 0 0 0 ?in the monthly advertising budget, will double...
-
Sales Weaver Company Income Statement For This Year Ended December 31 Cost of goods sold Gross margin Selling and administrative expenses Net operating income Nonoperating items: Gain on sale of...
-
1. Reflect on the importance of the availability of electricity to a country's level of economic development. What is the impact of electricity on the lives of people in Kenya? How has it helped the...

Study smarter with the SolutionInn App


