Consider the equilibrium between copper and its oxide: The Gibbs energy of formation of Cu2O is given
Question:
Consider the equilibrium between copper and its oxide: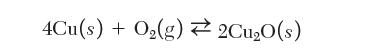
The Gibbs energy of formation of Cu2O is given by![]()
Make a plot of pO2 vs. T, illustrating where Cu is stable and where Cu2O is stable in the temperature range of 300 K to 1300 K.
Transcribed Image Text:
4Cu(s) + O(g) 2Cu0(s)
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The equilibrium pressure of H, over solid uranium and uranium hydride, UH3' at 500 K is 139 Pa. Calculate the standard Gibbs energy of formation ofUH3 (s) at 500 K.
-
The Soviet Venera VII probe, which reached Venus on December 15, 1970, found the following conditions on the planet surface: Interferometric measurements indicate that there is only a 10 to 20 K...
-
The following data represents the number of Grand Jury indictments for Gloucester County, New Jersey for a sample of 11 weeks selected from July 2010 through June 2011 as reproduced from that...
-
Suppose that the Medicare rate of hospital reimbursement is reduced. Explain why the costs may not be shifted to other patients in the short run.
-
How might the concept of Crowd funding impact the traditional donor relationship development process? What implications might this have on the organization's fundraising ability in the long-run?
-
Set up the triple integral of an arbitrary continuous function f (x, y, z) in cylindrical or spherical coordinates over the solid shown. ZA .
-
Number of classes: 5 Data set: Strengths (in parts per thousands) of 24 acids Construct a frequency distribution and a frequency histogram for the data set using the indicated number of classes....
-
The Roniger Company produces two products: bed mattresses and box springs. A prior contract requires that the firm produce at least 30 mattresses or box springs, in any combination. In addition,...
-
Design a Moore FSM to detect if an input sequence is divisible by 5. Each cycle a new bit comes as input towards LSB (e.g. if the current sequence is '1011' and the input is '0', the new sequence is...
-
Consider the reaction of CrCl2 with H2 to form solid Cr as follows: At Answer the following questions: (a) From these data, estimate the enthalpy of reaction. (b) In an attempt to increase the extent...
-
Hydrogen cyanide can be manufactured by reaction of acetylene and nitrogen: Calculate the equilibrium composition at 800 K and 1 bar. CH + N 2HCN
-
The concentration of the alkane C20H42 (FM 282.55) in a particular sample of rainwater is 0.2 ppb. Assume that the density of rainwater is close to 1.00 g/mL and find the molar concentration of...
-
When developing pro-forma financial statements, what reconciliation of accounts is Not Necessary?
-
Anticipate an extended discussion on how to best leverage our financial capital to create competitive advantage during our next board meeting. With this in mind, I'd like to better understand the...
-
Sample speeches and lectures with audio and video clips Brochures or programs from exhibits and shows Proposals and other written documents prepared on the job Names and contact information for...
-
A patient in the post-surgical period is received in the recovery room. Which part of the estimate should be a priority for the nursing staff? A patient in the post-surgical period is received in the...
-
What is an explanation for the amount of the Caldwells' refund or balance due?
-
When asked about his previous salary in a job interview, Jeremy boosts his salary a bit. He reasons that he was about to get a raise, and he also felt that he deserved to be paid more than he was...
-
Chloroplasts are illuminated until the levels of the Calvin cycle intermediates reach a steady state. The light is then turned off. How does the level of RuBP vary after this point?
-
The total cross-sections for reactions between alkali metal atoms and halogen molecules are given in the table below (R.D. Levine and R.B. Bernstein, Molecular reaction dynamics, Clarendon Press,...
-
For the thermal decomposition Of F2O by the reaction 2 F2O (g) 2 Fl (g) + 0z (g),) Czarnowski and H) Schumacher (Chem. Phys. Lett. 17, 235 (1972)) have suggested the following mechanism: (a) Using...
-
One of the most historically significant studies of chemical reaction rates was that by M. Bodenstein (z. physik. Chem. 29,295 (1899)) of the gas-phase reaction 2 Hl(g) -t H2(g) + I2(g) and its...
-
Hazard identification in the workplace is the responsibility of what ? Explain
-
A digital clock with a time reading 09:46. What is the absolute uncertainty of the measurement? answer: 0.5 minutes
-
Consider the following macro model where a, m are positive parame- ters, and t is the given average tax rate. Y C = C+ Go+N 100+ a(Y - tY) N = 50-mY (i) Write the system in matrix form AX = B, where...

Study smarter with the SolutionInn App


