Table 6.1 presents data for the enthalpy of solution, for nitric acid in water at 18C. Find
Question:
Table 6.1 presents data for the enthalpy of solution, 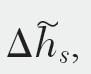 for nitric acid in water at 18°C. Find the corresponding values for the enthalpy of mixing vs. mole fraction of solute.
for nitric acid in water at 18°C. Find the corresponding values for the enthalpy of mixing vs. mole fraction of solute.
Transcribed Image Text:
Ahs, S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Let the solute HNO3 be species 1 and the solvent HO be species 2 The enthalpy of solution can be wri...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Which of the following would produce a materials price variance? abreakage of materials in production. ban excess quantity of materials used can excess number of direct labor-hours worked in...
-
Security Operations Centers (SOCs) around the world offer a range of cyber security services to organizations with the intention to protect information. They form the backbone of I.T. and security...
-
The molar integral heat of solution s H is defined as the change in enthalpy that results when 1 mole of solute (component 1) is isothermally mixed with N 2 moles of solvent (component 2) and is...
-
The hot dog cooker described in the chapter heats hot dogs by connecting them to 120 V household electricity. A typical hot dog has a mass of 60 g and a resistance of 150 N. How long will it take...
-
Critically analyze current European and United States industry standards or recommendations for any Information Technology (IT) area or subarea (e.g., intrusion detection, data recovery, data...
-
How useful is a financial plan when it is based on assumptions of the future and we are confident that these assumptions are not going to be 100 percent correct? Does it make more sense for the...
-
Assuming factorability of the complex degree of coherence of polarized thermal light, demonstrate that when the photodetector area is much larger than the coherence area of the incident light, the...
-
Perform assignment 7 using the law from your state. Assignment 7 In the following exercise, the assignment is to prepare a trial court brief. The assignment contains the memo from the supervising...
-
Describe the 'Big Five' factors as they relate to HRM (do not just list them). Which seem more related to managerial positions? Why?
-
Develop an expression for the entropy change of mixing for a binary ideal gas mixture.
-
An insulated pistoncylinder assembly contains two compartments divided by a partition. As shown in Figure E6.9A, the top compartment initially contains 2 moles of pure liquid 1, whereas the bottom...
-
What are the special points to which an auditor should direct his attention for ascertaining the adequacy of provision for bad and doubtful debts in the context of proper valuation of sundry debtors?
-
Set your calculator to 4 or more decimal places. 1. You invest $7,000 at the end of each year for 10 years. The investment value at the end of 10 years is $106,351. What is the annual rate of return?...
-
Investment income related to Assets Whose Use Is Limited amounted to $15,000 for St, Mary's Hospital, is a not-for-profit business-oriented hospital. The hospital's board designated the money for...
-
Explain the role of Explanations of Benefits (EOBs) and Medicare Summary Notices (MSNs) in educating consumers.
-
1) Terps Cookies has been experiencing dramatic growth. It had sales of $15,000 in 2014 and $18,000 in 2015. Sales are projected to grow at a rate of 8% in 2016 but then slow to 5% after 4 years....
-
Jane is saving money so that she will have $9,000 in 6 years. She plans to make payments at the end of each quarter into an account paying interest at an annual nominal rate of 6.2% compounded...
-
Identify a process used by a majority of business professionals in resolving problems with disappointed customers.
-
Design a circuit which negative the content of any register and store it in the same register.
-
Explicit expressions for hydrogenic orbitals are given in Tables 10.1 and 9.3. (a) Verify both that the 3px orbital is normalized (to I) and that 3px and 3dxy are mutually orthogonal. (b) Determine...
-
Show that l, and 12 both commute with the Hamiltonian for a hydrogen atom. What is the significance of this result?
-
Some atomic properties depend on the average value of 1/r rather than the average value of r itself. Evaluate the expectation value of 1/, for? (a) A hydrogen 15 orbital, (b) A hydrogenic 25 orbital,...
-
What would be two accounting practices related to tax documentation for individuals?
-
A simply supported beam is subjected to a uniform service dead load of 1.0 kips/ft (including the weight of the beam), a uniform service live load of 2.0 kips/ft, and a concentrated service dead load...
-
What can assist when defining acceptable level of risk by identifying community members' motivations that drive their decision-making. Explain

Study smarter with the SolutionInn App


