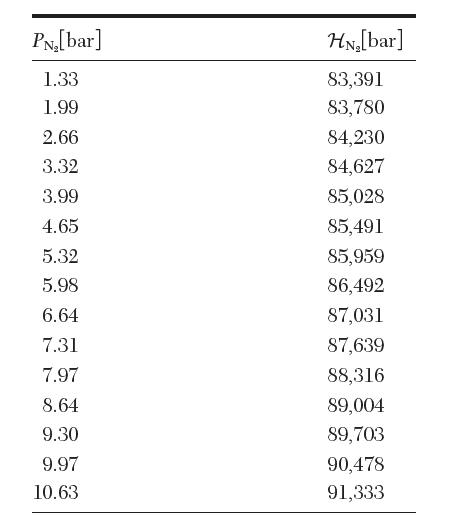
The following data are available for the Henrys law constant of N2 in H2O at 19.4C. From
Question:
The following data are available for the Henry’s law constant of N2 in H2O at 19.4°C. From these data, estimate![]()
Transcribed Image Text:
00: 8 N*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Compute the CLV metric and then apply it in a simple breakeven analysis. The data below show the results of a Starbucks segmentation study. Each column shows the values for a typical customer in each...
-
The following data are available for 2012, regarding the inventory of two companies: Compute inventory turnover and number of days sales in inventory for both companies. Which company is handling its...
-
The following data are available for Haul-It-Away Truckers: 1. Compute the predetermined overhead rate for each of the two years, if based on (a) Direct labor hours, (b) Number of moving jobs, and...
-
Determine whether each function is one-to-one. If it is, find the inverse. f(x) = 4x - 1
-
From the following account information, prepare an income statement cost of goods sold section in proper form: Freight-In, $400; Merchandise Inventory, 12/31/17, $9,500; Purchases Discounts, $800;...
-
Use the figures to calculate the left and right Riemann sums for f on the given interval and for the given value of n. on [1,5]; n = 4 f(x) f(x) = 1/x f(x) = 1/x 1 1 2 3 4 2 3 4.
-
Using the result from Problem 3.9, determine the effective Young's modulus, \(E_{x}\), for the RVE shown in Figure 3.13. In Figure 3.13, the reinforcing particle has a square cross section and is...
-
A furniture factory makes two types of wooden tables, large and small. Small tables are made in batches of 100, and large tables are made in batches of 50. A batch includes a fixed setup time for the...
-
Consider the soil profile shown in Figure E3-2. The moist unit weight of the top sand layer is 20.0 kN/m. Capillary rise is present above the groundwater table. The saturated unit weights of clay 1...
-
The following data are available for the Henrys law constant of O2 in benzene. From these data, estimate Ho, - ho:
-
A binary mixture of carbon dioxide and water exists in vaporliquid equilibrium at 343.15 K and 1 bar. The solubility of CO2 in the liquid has been measured as xCO2 = 0.000255. What is the Henrys law...
-
A function is given by a table of values, a graph, a formula, or a verbal description. Determine whether it is one-to-one.
-
Assume you are a machine learning lead in a software development company. Management has tasked you to improve the customer contact center using Machine Leaning and language models, for example, Chat...
-
Consider the following utility function, u (, 2) = min [ 2, where y>0 1 (a) [20 points] Derive the Marshallian demand functions. (Show detailed steps of your derivation.) Does the Marshallian demand...
-
Consider the following model of moral hazard: A risk-neutral principal offers a contract to a risk-averse (uA(z) = ) agent. There are two possible levels of output q {qL9H}. There are two possible...
-
This assignment asks you to use the techniques that you have learned in this course to evaluate a potential investment opportunity. Specifically, it asks you to use the data provided with this...
-
With you being the CEO of Kerr's Bakery & more Located in Kingston, Jamaica Baked products of cakes, pudding and pastries, Create the following for your company: 1. Rational and Objective 2.Target...
-
By focusing on the Pay Less part of its slogan, has Target pursued the best strategy? Why or why not? When you hear the term discount retail, two names that usually come to mind: Walmart and Target....
-
Before the 1973 oil embargo and subsequent increases in the price of crude oil, gasoline usage in the United States had grown at a seasonally adjusted rate of 0.57 percent per month, with a standard...
-
Give the structure of the major alkene formed when the hydroxide of each of the following quaternary ammonium ions is heated.
-
Outline syntheses of each of the following from aniline and any necessary organic or inorganic reagents: (a) p-Nitroaniline (c) p-Aminoacetanilide (b) 2, 4-Dinitroaniline
-
N-Nitroso amines are stabilized by electron delocalization. Write the two most stable resonance forms of N-nitrosodimethylamine, (CH3)2NNO.
-
What are the key principles underlying the design and operation of multiphase reactors for the synthesis of fine chemicals, and how do they impact overall reaction kinetics and selectivity? Explain
-
Find three scholarly articles that relate to a personality disorder of your choice (List is below) Summarize the findings per article. Paranoid Personality Disorder (Cluster A) Schizoid Personality...
-
Accounting and financial reporting for governmental entities, including fund accounting, special funds, and government wide financial statements. Retrieve a recent Comprehensive Annual Financial...

Study smarter with the SolutionInn App


