The Gibbs energy of a binary mixture is given by the following expression: where R is the
Question:
The Gibbs energy of a binary mixture is given by the following expression: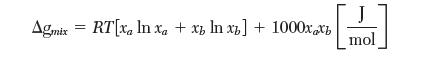
where R is the ideal gas constant and T is in [K]. For a mixture at 298 K with the following mole fractions of a, calculate Ga - ga.
(a) xa = 1
(b) xa = 0.4
(c) xa = 0 (a in infinite dilution)
Transcribed Image Text:
Agmix = RT[xa In xa + xz ln xz] + 1000xxb mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The Gibbs energy of a binary mixture of species a and species b at 300 K and 10 bar is given by the following expression: (a) For a system containing 1 mole of species a and 4 moles of species b, fi...
-
Suppose the (1) + (2) system exhibits liquid-liquid immiscibility. Suppose we are at a state where G 1 /RT = 0.1 and G 2 /RT = 0.3. The Gibbs energy of mixing quantifies the Gibbs energy of the...
-
(a) The molar Gibbs energy of mixing (per mole of superficial solution) for a liquid binary system expressed extensively, this becomes Introduce the concepts of chemical theory into Eqn. 19.131 to...
-
Children out of School in America is a report on delinquency of school-age children prepared by the Childrens Defense Fund (CDF), a government-sponsored organization. Consider the following three...
-
How do the literatures of America's later Colonial and Revolutionary eras reflect contemporary political, economic, and cultural changes?" How do those works of literature written during the latter...
-
Examine the role wireless networks are having in the developing world. Why are some places bypassing LANs and physical cabling altogether and moving to a wireless system architecture? What are the...
-
Starting with Eq. (4.1-10), show that if \(\Delta v \ll \bar{v}\) and \(r \ll 2 c / \Delta v\) for all \(P_{1}\), then \[ \mathbf{u}\left(P_{0}, t ight) \approx \iint_{\Sigma} \frac{e^{j 2 \pi(r /...
-
On November 1, 2015, Norwood borrows $200,000 cash from a bank by signing a five-year installment note bearing 8% interest. The note requires equal total payments each year on October 31. Required 1....
-
2. a) Illustrate how BUILD-MAX-HEAP (A) works on the array A = {2, 9, 6, 8, 11, 4, 5, 3, 1, 7}. Each step is the result of one call to the MAX-HEAPIFY function. Show both the array view and the tree...
-
The molar enthalpy of a binary liquid mixture of species 1 and 2 is given by: where T is the temperature in [K] (a) What is the enthalpy of mixing, Hmix in [J], of a mixture with 2 mol of 1 and 3 mol...
-
Enthalpies of mixing for binary mixtures of cadmium (Cd) and tin (Sn) have been fi t to the following equation at 500C: where, XCd and XSn are the cadmium and tin mole fractions, respectively....
-
In triangle ABC below, the measure of angle A is 20 and the measure of angle B is 3 times larger than the measure of angle C. What is the measure of angle B? A. 40 B. 60 C. 80 D. 120 E. 160 B
-
How do intrinsic and extrinsic motivators interact within the framework of self-determination theory to shape individuals' pursuit of complex goals ?
-
Describe Warren Buffet and Charlie Munger's reaction to cryptocurrency/markets. Does their age distort their understanding and opinion of this market?
-
In a group of 25 students at lunch, 10 prefer ketchup on their hamburger, 10 prefer mustard on their hamburger, and 5 like both ketchup and mustard. What is the probability that a student selected...
-
what ways do achievement-oriented individuals leverage goal-setting theory to optimize their performance and sustain high levels of motivation over extended periods ?
-
To conduct this manufacturing they import certain electronic components from countries like Japan and South Korea. The market for the manufactured tools are the U.S.A, Australia, Canada and United...
-
Consumer Claim: The Check in the Mail Is a Bill Houston chiropractor Brett Downey cashed a $2.50 check last December from Yellow Pages Inc. of Anaheim, California. That is when his troubles began. He...
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
In a study of the rotational spectrum of the linear Fe CO radical, K. Tanaka, M. Shirasaka, and T. Tanaka (J. Chem. Phys. 106,6820 (1997)) report the following J+ I (,- J transitions: J 24 25 26 27...
-
Predict the shape of the nitronium ion, NO from its Lewis structure and the VSEPR model. It has one Raman active vibrational mode at 1400 cm-1, two strong IR active modes at 2360 and 540 cm-1, and...
-
The HCI molecule is quite well described by the Morse potential with De = 5.33 eV, V = 2989.7 cm-1, and XV = 52.05 cm-I. Assuming that the potential is unchanged on deuteration, predict the...
-
What is Archaeologists working in cultural resource management?
-
Janie graduates from high school in 2023 and enrolls in college in the fall. Her parents (who file a joint return) pay $10,975 for her tuition and fees. If required, round your computations to the...
-
19-year-old white male found by his roommate in a "sluggish" state following the ingestion of 10 sleeping pills (Sominex) and one bottle of whiskey. Recently has been giving away his possessions and...

Study smarter with the SolutionInn App


