Question: Using data from Table 6.1, fi nd the partial molar enthalpy of water in a mixture of ethanol (1) and water (2) at 25C with
Using data from Table 6.1, fi nd the partial molar enthalpy of water in a mixture of ethanol (1) and water (2) at 25°C with x1 = 0.33. Use the same reference state as used in the steam tables.
Steam tables
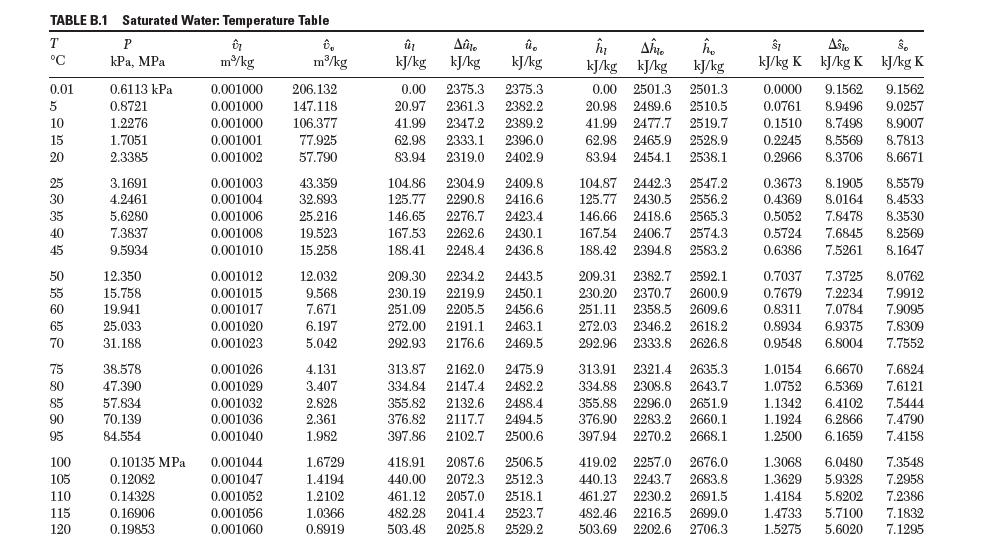
TABLE 6.1 Enthalpy of Solution of Different Species in Water at 25C Ah, J mol solute n NaOH CH5OH ZnCl NaCl HPO4 CH4O NH 2,510 628 -29,539 -812 669 -32,049 -1,799 -2,787 586 -32,761 -28,886 -33,263 -34,430 [mol HO] HNO HSO4 HC -13,113 -31,087 -26,225 -20,083-44,936 -48,819 -45,886 -837 -24,301 -52,007 -56,852 -46,798 -4,184 -26,978 -57,070 -61,204 -47,187 -6,276 -3,757 -28,870 -28,727 -61,045 -64,049 -47,384 -7,531 377 -33,598 -37,761 -4,694 -32,677 -31,840 -70,040 -69,488 -47,719 -9,874 -209 -34,267 -42,501 -7,364-40,083 1,941 -32,669 -74,517 -71,777 -47,840 -10,962 -628 -34,434 -42,865 -8,866-46,191 2,671 -32,744 -76,358 -73,729 -47,949 -11,966 -1,130 -34,518 -42,526 -9,975 -55,020 3,732 -32,748 -76,986 -73,848 -48,057 -12,468 -1,276 -34,560 -42,334 -10,268 -61,086 4,109 -33,338 -99,203 -75,189 -60,501 -1,435 -34,644 -42,869 -71,463 3,801 1 2 3 4 5 10 20 50 100 00 HF
Step by Step Solution
3.47 Rating (154 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


