Consider the isothermal mixing of 20% solute 1 by weight and 80% water, 2, at 25C. What
Question:
Consider the isothermal mixing of 20% solute 1 by weight and 80% water, 2, at 25°C. What is the heat transferred for the following mixtures?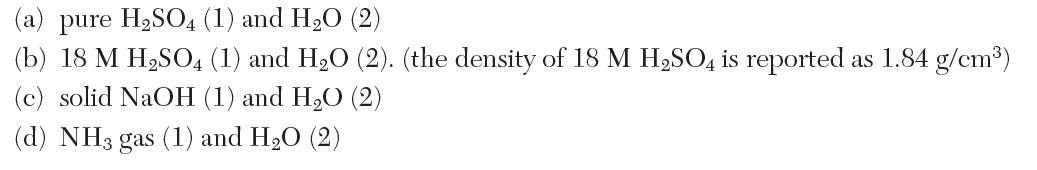
Transcribed Image Text:
(a) pure HSO4 (1) and HO (2) (b) 18 M HSO4 (1) and HO (2). (the density of 18 M HSO4 is reported as 1.84 g/cm) (c) solid NaOH (1) and HO (2) (d) NH3 gas (1) and HO (2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Briefly describe ASCII and Unicode and draw attention to any relationship between them. [3 marks] (b) Briefly explain what a Reader is in the context of reading characters from data. [3 marks] A...
-
If an investor holds a credit-linked note and the credit event does not occur, the investor receives: A. All promised cash flows as scheduled. B. All coupon payments as scheduled but not the par...
-
Review the data model 1- Provide definitions of each of the following components: a- Entities b- Attributes c- Relationships d- Primary keys Identify each of the components from the data model above:...
-
Carol Lorenz was employed by Tyson Foods, Inc. in Cherokee, Iowa, from October 1985 until December 6, 2012, when her employment was terminated. She was 62 years old at the time of her discharge....
-
The sun subtends an angle of about \(32 \mathrm{~min}\) of arc ( 0.0093 radians) on Earth. Assuming a mean wavelength of \(550 \mathrm{~nm}\), calculate the coherence diameter and coherence area of...
-
Jerry Rice and Grain Stores has $4,780,000 in yearly sales. The firm earns 4.5 percent on each dollar of sales and turns over its assets 2.7 times per year. It has $123,000 in current liabilities and...
-
A+ Rental Cars corporate office offers each franchise location $20,000 each month to spend on local advertising. Some franchises take advantage of this offer, while others do not. We have gone back...
-
The following data are available for a binary mixture of ethanol and water at 20C: (a) Make a plot of the partial molar volumes of ethanol and water vs. mole fraction ethanol. (b) What is vmix for an...
-
Using data from Table 6.1, fi nd the partial molar enthalpy of water in a mixture of ethanol (1) and water (2) at 25C with x1 = 0.33. Use the same reference state as used in the steam tables. Steam...
-
In horizontal analysis, each item is expressed as a percentage of the: (a) net income amount. (b) stockholders equity amount. (c) total assets amount. (d) base year amount.
-
Establish your organization. This budget will be at the DEPARTMENT (EX. FIRE DEPARTMENT) level. What is your unit of service? What is your staffing mix - hours - shifts - skill level? Tangible and...
-
You are an audit team leader overseeing the activity of a trainee auditor. Your team has just completed a surveillance audit and is assembling the final report. The trainee has not contributed to a...
-
You have been hired by Kathy's Craft Supplies as a bookkeeper. You are reviewing the general journal transactions from the previous period. You immediately recognize that Kathy uses an accounts...
-
Adams, Inc. sells widgets that come with an unconditional five-year warranty. According to Adams' best estimates, 7.8% of all units sold will require repair or replacement under that warranty, and...
-
Kathy's Craft Supplies uses an accounts payable ledger. What would Kathy look for to verify that a transaction has been posted to the accounts payable ledger?
-
Document for Analysis: Poor Action Request for Internship Your Task. Analyze the poorly written persuasive request. List its weaknesses. If your instructor directs, revise the letter. Add any...
-
San Carlos Bank and Trust Company uses a credit-scoring system to evaluate most consumer loans that amount to more than $2,500. The key factors used in its scoring system are found at the conclusion...
-
There is some indication that other hydrogen ring compounds and ions in addition to H3 and D3 species may play a role in interstellar chemistry. According to J.S. Wright and G.A. DiLabio (J. Phys....
-
Molecular orbital calculations may be used to predict trends in the standard potentials of conjugated molecules, such as the quinones and flavins that are involved in biological electron transfer...
-
Explain how a molecule is assigned to a point group.
-
Find an analytic function whope real part is u=e" cosy.
-
What generally describe(s) the activity of planning, organizing, and controlling resources to achieve specific goals?
-
Find an analytic function whose read part 98 x-ty. .2

Study smarter with the SolutionInn App


