While returning to your dorm late last night with a hot cup of coffee, the heat overcomes
Question:
While returning to your dorm late last night with a hot cup of coffee, the heat overcomes you and, much to your chagrin, you drop the paper cup, spilling its entire contents. As you had just spent your laundry money, this is somewhat upsetting, especially since you still have a good deal of thermo left to study and your last clean pair of pants are now covered with coffee.
You yearn for the old days of polystyrene (Styrofoam) cups, which never got hot. Being an ambitious student (and looking for a distraction), you decide to come up with a process to recycle polystyrene (Styrofoam) so that environmental concerns will no longer keep the coffee shop from using this very good insulating material.
After several hours, you have come up with what you think is a very reasonable process (you cannot wait to call the patent attorney!) and have just a few fi nal issues to resolve. In the purifi cation process, you believe you have reduced the polystyrene to its monomer, styrene, shown below: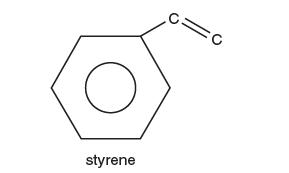
In this case, the reactor would consist of 100 moles of styrene in a volume of 30 L at a pressure of 10 bar. You are concerned that the temperature is beyond the limit for the decomposition of styrene, 289°C.
Since you are studying for the thermo exam, and have just gotten to the van der Waals equation, you want to decide whether this would be a good equation of state to use.
(a) What deviations from ideality would you expect at these reaction conditions? In order of importance, list the types of intermolecular forces you think contribute to nonideality. Is the van der Waals equation appropriate? Explain.
(b) Your search for experimental values for the van der Waals constants, a and b, is futile; you do, however, fi nd values for the critical constants for styrene: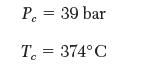
Calculate the temperature of the reactor, using the van der Waals equation. Will the styrene decompose?
(c) Your classmate, who’s taking a polymers class, says the polystyrene probably has not reduced to a monomer but still exists as a reduced polymer chain, perhaps fi ve monomers long: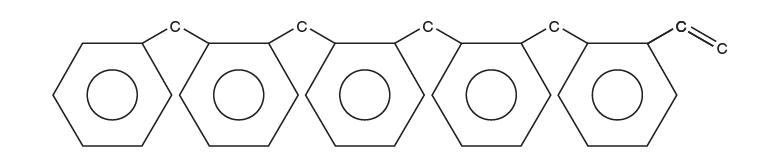
Using only the information above, what are reasonable values for the van der Waals constants, a and b, of this reduced polymer chain? Explain.
(d) Calculate the temperature in the reactor at the same reactor volume and pressure and initial Styrofoam mass as for part (b), except where you have a fi ve-unit polymer instead of the monomer.
Explain the difference in value to that calculated in part (b). Will decomposition occur (assume around 289°C)?
Step by Step Answer:






