Your coworker has scribbled down the saturation pressures for a pure species from the solid (sublimation) and
Question:
Your coworker has scribbled down the saturation pressures for a pure species from the solid (sublimation) and liquid (evaporation) as follows: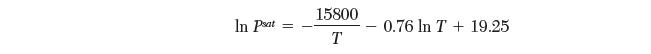
and,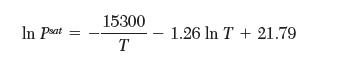
However, in his haste, he forgot to note which equation was for sublimation and which was for evaporation. Please help your coworker by determining the correct matches. Explain your reasoning.
Transcribed Image Text:
In Pat 15800 T 0.76 In T+ 19.25
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Northern Virginia Community College HOW MUCH FINANCIAL RISK SHOULD YOU TAKE? Mark D. D'Antonio Nova Southeastern University FORT LAUDERDALE, FLORIDA, U.S.A. Abstract A successful retirement...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Following procedures similar to those for the homogeneous problem (see Section 8.4.1), develop the following stress field for a pressurized hole in an infinite nonhomogeneous medium with moduli...
-
Regarding government budgets, explain the incrementalism model (championed by Lindblom). How does it change with the level of government? How is it affected by a divided government? Is it an...
-
Imagine that two countries, Richland and Poorland, can produce just two goods, computers and coal. Assume that for a given amount of land and capital, the output of these two products requires the...
-
Readings taken with this particular model yielded 50 mV/A. Based on this sensitivity, is the motor upon which the following readings were recorded in trouble? -42 42 -78 -114 -150 130 80 12 102 66 30...
-
Ren Alverez knew she was in over her head soon after she took the job. Even so, the opportunity for promotion comes along rarely and she believed that she would grow into it. Ms. Alverez is the cost...
-
Are way paying men and women equally? What is the p-value for the difference in Monthly Income by gender?
-
(a) A pure fl uid shows the following s vs. T behavior. Draw schematically how the chemical potential would change with temperature. (b) A pure substance shows the following v vs. P behavior at...
-
A well-insulated tank with a valve at the top contains saturated water at 5 MPa. The quality of the water is 0.1. (a) What is the ratio of the liquid volume to the vapor volume? (b) The valve is...
-
A game of chance based on a spinning wheel is available that pays \(n\) times money bet in the case of a win and nothing in the case of a loss. A gambler has developed a device by which he may...
-
Explain why MNCs such as Coca Cola and PepsiCo, Inc., still have numerous opportunities for international expansion. Explain briefly
-
Review the meaning of the concepts or terms given in Key Terms and Concepts. Distinguish between a production department and a service department. Distinguish between a direct cost and an indirect...
-
What would you like the members of your learning community to know about your global health interests, perspectives, and life experiences? How will you prepare to listen and learn from others? Why...
-
3. A large fraction of deposits in the banking system is not insured, i.e., it is above the 250K thresholds. In this question, we will analyze the households' banking accounts. a) FDIC is the...
-
Meredith is the named insured on a Personal Auto Policy that provides the following coverages: 1974 AMC Gremlin 2015 Honda Liability Liability Medical Payments Medical Payments UM $250 ded. OTC...
-
Outline the writing plan for a message that requests routine information or action.
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
Treat carbon monoxide as a perfect gas and apply equilibrium statistical thermodynamics to the study of its properties, as specified below, in the temperature range 100-1000 K at 1 bar. V = 2169.8...
-
The exchange of deuterium between acid and water is an important type of equilibrium, and we can examine it using spectroscopic data on the molecules. Calculate the equilibrium constant at (a) 298 K...
-
Suppose that an intermolecular potential has a hard-sphere core of radius 'I and a shallow attractive well of uniform depth E out to a distance '2' Show, by using eqn 17.42 and the condition E kT,...
-
Intro Button Snowboards is a small, independent manufacturer of round snowboards. Each board is sold for $410. Fixed operating costs are $424,000 per year and variable costs are $320 per board.
-
Question 1: Undertake online research and, based on your knowledge and experience, what are the key factors that determine the capital structure of a company? At least three suggestions with a brief...
-
In November 2010, Hammer Technologies issued a series of 7%, 30-year semi-annual coupon bonds. Interest rates decreased in subsequent years and by November 2020, the price of the bonds had increased...

Study smarter with the SolutionInn App


