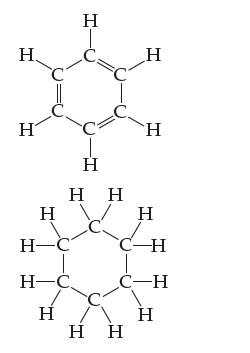
A hexagonal molecule has angles of 120 inside the ring and is flat. The following two molecules
Question:
A hexagonal molecule has angles of 120° inside the ring and is flat. The following two molecules are often drawn as flat hexagons, but only one is truly flat. Which one is it and why?
Transcribed Image Text:
Η. H Η H-C Η Η H¬C Η Ι Η Η Η H Ἡ Η C-H Η C-H Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The molecule on the left is truly flat because it has angles of 120 inside the ringwhich is consiste...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
A uniformly charged thin ring has radius 15.0 cm and total charge +24.0 nC. An electron is placed on the ring's axis a distance 30.0 cm from the center of the ring and is constrained to stay on the...
-
A container of weight W is suspended from ring A. Cable BAC passes through the ring and is attached to fixed supports at B and C. Two forces P= Pi and Q= Qk are applied to the ring to maintain the...
-
Find the derivative of the vector function r(t) = e^t^2 i-j + In(1+3t)k.
-
Go to the text Web site at www.cengage.com/accounting/vanderbeck and click on the link to kaizen, from Wikipedia, the free encyclopedia. After reading the entry, answer the following questions: 1....
-
Intex Potatoes (IP) operates at capacity and processes potatoes into potato cuts at its highly automated Pocatello plant. It sells potatoes to the retail consumer market and to the institutional...
-
If you had been treated with respect, this would never have happened, would it?
-
Comprehensive variance analysis. Sol Electronics, a fast-growing electronic device producer, uses a standard costing system, with standards set at the beginning of each year. In the second quarter of...
-
A 120-room building has 14 1-bedroom units renting monthly at $150.00. 12 2-bedroom units at $200.00, and 8 3-bedroom units at $250.00. The gross annual rental would be?
-
Consider CH 3 + and CH 3 . Using these as examples, explain why it is necessary to draw a correct Lewis dot diagram before trying to predict the shape of a molecule. Use the concept of steric number...
-
Consider the following molecule. The diagram shows how the atoms are connected, but it is not a complete dot diagram. (a) Complete the dot diagram. (b) Redraw the molecule showing its three...
-
Peter Ltd produces one product, the Baz. The standard direct material cost of each unit was as follows: 2.5 kilos of material at $4.80 per kilo = $12 per unit. During February 2015, the company...
-
Yem Company manufactures luggage sets. Yem sells its luggage sets to department stores. Yem expects to sell 2,050 luggage sets for $320 each in January and 2,200 luggage sets for $320 each in...
-
Mark owns a machine shop. In reviewing the shops utility bills for the past 12 months, he found that the highest bill of $2,400 occurred in August when the machines worked 1,000 machine hours. The...
-
Refer to the Packers schedule of cash receipts from customers that you prepared in Short Exercise S22A-17. Now assume that Packerss sales are collected as follows: 50% in the month of the sale 20% in...
-
What is the purpose of variable overhead cost variance?
-
What does profitability mean with regard to performance evaluation in investment centers?
-
Five different analysts have submitted valuations for a private technology firm that is the subject of a possible acquisition. The valuations are as follows: Analyst Valuation 1 ......... $26,331,000...
-
Four GWU students have been selected to taste food sold by 3 different food trucks labeled as food truck A, B and C on H & 22nd Streets every Monday for 3-weeks. For each student, food trucks are...
-
How would you use IR spectroscopy to distinguish between the following pairs of compounds? (a) (b) N.
-
The amplitude of a standing wave function representing a moving particle can change from positive to negative values in the domain (0, a) over which the wave function is defined. It must therefore...
-
According to the 3rd postulate, in any single measurement of the total energy, the only values that will ever be measured are the eigenvalues of the total energy operator. Apart from the discrete...
-
Book Value versus Market Value Masterson, Inc., has 4.1 million shares of common stock outstanding. The current share price is $84, and the book value per share is $11. The company also has two bond...
-
The most recent financial statements for Cardinal, Inc., are shown here: Income Statement Balance Sheet Sales $ 33,000 Assets $ 77,200 Debt $ 40,200 Costs 18,650 Equity 37,000 Taxable income $ 14,350...
-
How do genetic and epigenetic factors influence metabolic phenotypes and contribute to metabolic heterogeneity among individuals, shaping susceptibility to metabolic diseases and therapeutic...

Study smarter with the SolutionInn App


