(a) Rank these molecules in order of increasing boiling point: (b) State your reason for the order...
Question:
(a) Rank these molecules in order of increasing boiling point:
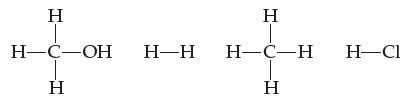
(b) State your reason for the order you chose in part (a).
Transcribed Image Text:
H H-C-OH H H-H H H-C-H H H-Cl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
To rank the molecules in order of increasing boiling point we consider the intermolecular forces pre...View the full answer

Answered By

Sufiyan Ahmed Tariq
I am a Chartered Accountant and an Associate Public & Finance Accountant. I also hold a bachelors of Commerce degree. I have over 8 years of experience in accounting, finance and auditing. Through out my career, I have worked with many leading multinational organisation.
I have helped a number of students in studies by teaching them key concepts of subjects like accounting, finance, corporate law and auditing. I help students understanding the complex situation by providing them daily life examples.
I can help you in the following subject / areas:
a) Accounting;
b) Finance;
c) Commerce;
d) Auditing; and
e) Corporate Law.
4.90+
7+ Reviews
17+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
(a) Rank these molecules in order of increasing boiling point: (b) State your reason for the order you chose in part (a). Br Br-C-Br Br Br Br H H-C-H H CI-CI
-
Rank the following molecules in order of increasing boiling point (without looking up the real values!): (a) 3-methylheptane; (b) Octane; (c) 2,4-dimethylhexane; (d) 2,2,4-trimethylpentane.
-
Rank the following isomers in order of increasing boiling point, and explain the reasons for your order of ranking. OCH2CH2OH CH C 2-(vinyloxy ethanol OCH2CH CH,CH CHC-OH ethyl acetate butyric acid
-
What is food processing ? Different types of food processing methods Briefly explained
-
Discuss how technology has changed jobs in an organization where you have worked. What are some HR responses to those changes?
-
In Problem 2, assume that the inflows are uncertain but normally distributed with standard deviations of $1000, $1500, $2000, and $3500, respectively. Find the mean forecast NPV using Crystal Ball....
-
Estimate the overall odds ratio of the set of tables in Problem3.6 and test whether the odds ratios are the same across the tables. Problem3.6 is: 3.6 Use the DOS data to test whether there is gender...
-
Keep Cool Industries, Inc., manufactures fans for personal use. Department 70 is responsible for assembling the fan. Department 71 packages them for shipment. Keep Cool recently produced 10,000 fans...
-
Contemporary psychoanalysis has undergone a paradigm shift from drive reduction to the relational model. The most fundamental difference between relational and classical forms of psychoanalysis is...
-
Which compounds would you expect to experience only London forces? Explain your choices. (a) Carbon tetrabromide, CBr 4 (b) Methyl bromide, CH 3 Br (c) Phosphorous tribromide, PBr 3 (d) Boron...
-
Show how two CH 2 Cl 2 molecules in the liquid phase are oriented with respect to each other and explain why the molecules align this way.
-
Discuss how the aggregate planning model could be extended to handle a company that produces several products on several types of machines. What information would you need to model this type of...
-
Show the following transactions in the journals of L Buhinjak, initially assuming periodic inventory applies and then where perpetual inventory applies. 29 August 2022: Stock costing $1200 and tax...
-
J Leeson commenced business on 15 April 2022 with $5000 in the bank, GST receivable $350 and a computer valued at $3500. 20 April: Additional funds of $15 000 were injected by J Leeson into the...
-
On 1 March 2022, A Levstek commenced business with bank $10 000, GST receivable $4200, land $25 000, equipment $2000 and a motor vehicle $15 000. On 1 April 2022, A Levstek contributed further funds...
-
M Farrah purchased goods from D Michael for $25 905 ($23 550 + $2355 GST) on 15 April 2022. On 7 May a bill payable is signed, maturing on 28 November 2022. The bill is met on the due date. Show in...
-
On 18 February 2022, P Jackson sold stock to R Parramatta on credit for $3300 ($3000 + $300 GST); the cost of the goods sold was $1800. R Parramatta paid by cheque, which was banked on 27 February....
-
List causes that could lead to a production- volume variance?
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
What is an elementary chemical step, and how is one used in kinetics?
-
What is the difference between chemical and physical methods for studying chemical kinetics?
-
What are the fastest time scales on which chemical reactions can be investigated? Describe three experimental techniques for investigation of chemical reactions.
-
Air, at P-2 bar and v, -0.07 m/kg is heated at constant pressure until reaching the temperature T-104 C (state 2). The added heat is Q12-30 kJ/kg. Then undergoes an isothermal process and arrives at...
-
Remote teams yes, prove to add different issues of their own due to the nature of not being solely located in a FTF format for staff to work through any issues. But, at the same time, are these...
-
Why are dress code policies important to have in place in small companies?

Study smarter with the SolutionInn App


