Acetic acid, is very soluble in water because of the many hydrogen bonds that can form. Add
Question:
Acetic acid,
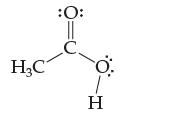
is very soluble in water because of the many hydrogen bonds that can form. Add water molecules to this drawing and show the hydrogen bonds possible.
Transcribed Image Text:
H₂C :O: С Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Acetic acid has two hydrogen bond donors a hydroxyl hydrogen and a methyl hydrogen and two hydrogen bond acceptors a carbonyl oxygen and a hydroxyl ox...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Many moderately large organic molecules containing basic nitrogen atoms are not very soluble in water as neutral molecules, but they are frequently much more soluble as their acid salts. Assuming...
-
In 2002, workers at the Swedish National Food Administration discovered that heated, carbohydrate-rich foods, such as french fries, potato chips, and bread, contain alarming levels (0.1 to 4 g/g) of...
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
Suppose that for a sample of n = 11 measurements, we find that n = 72 and s = 5. Assuming normality, compute confidence intervals for the population mean with the following levels of confidence: a....
-
Firms in a variety of industries have been recognized as being among the Most Admired Companies by Fortune magazine. Others have been highlighted as being the Best Companies to Work For by Fortune or...
-
List all the various ways that resources greatly increase scheduling complexity.
-
The price of a non-dividend paying stock is \($19\) and the price of a three-month European call option on the stock with a strike price of \($20\) is \($1.\) The risk-free rate is 4% per annum. What...
-
Krall Company uses a job order cost system. The following data summarize the operations related to production for June 2012, the first month of operations: a. Materials purchased on account,...
-
A vehicle of mass m = 1500kg travels on a circular curve of radius R = 1km at a speed of v = 10m/s. There is static friction between the road and the vehicle's tires. R Road with a circular curve of...
-
(a) Rank these molecules in order of increasing boiling point: (b) State your reason for the order you chose in part (a). Br Br-C-Br Br Br Br H H-C-H H CI-CI
-
(a) Name the strongest intermolecular force in CH 3 OH, CH 3 Cl, CH 3 CH 3 , and CH 3 CH 2 CH 3 . (b) Rank these molecules from lowest to highest boiling point.
-
Explain at least three types of projects in which large net cash flow changes may cause sign changes during the life of the project, thus indicating the possible presence of multiple ROR values.
-
C Kevin is having financial problems. He has not been receiving funds from some accounts receivable according to the trading terms and also has not been paying some credit suppliers according to...
-
Having checked the monthly accounts of Honey Suckle for account allocations, three errors were found that needed correction through the general journal. Subsidiary ledgers and control accounts are...
-
Show the following transactions in the two journals of S Alderson. Assume first that periodic inventory applies and then that perpetual inventory applies. 19 June 2022: Goods were sold to R...
-
Inventory was delivered to a customer on 30 June 2022 valued at $7579 ($6890 + $689 GST). However, there was a problem with the invoicing process and a tax invoice was not raised for the customer....
-
Show the following transactions in the two journals of P Kulevska. Assume that perpetual inventory applies. 5 October 2022: P Kulevska deposited $792 into her bank account. This was from a cash sale...
-
List causes that could lead to a variable overhead spending variance?
-
Which of the followingcarbocations is the least stable? CH3CH2 . CH3CHCH3 CH3 I . CH3C0 T CH3 IV. V. CH3 CH3CCH2 CH3
-
What is the steady-state approximation, and when is this approximation employed?
-
In a parallel reaction in which two products can be formed from the same reactant, what determines the extent to which one product will be formed over another?
-
What is the kinetic definition of equilibrium?
-
A house painting business had revenues of $16,500 and expenses of $9,500 last summer. There were no depreciation expenses However, the business reported the following changes in working capital:...
-
What is a good problem statement for high turnover rates in fast food?
-
Gluon Inc. is considering the purchase of a new high pressure glueball. It can purchase the glueball for $40,000 and sell its old low-pressure glueball, which is fully depreciated, for $6,000. The...

Study smarter with the SolutionInn App


