Actually, once in water, the amino acid glycine (see problem above) exists as a zwitterion, as shown
Question:
Actually, once in water, the amino acid glycine (see problem above) exists as a zwitterion, as shown below. A zwitterion is an overall electrically neutral species that nevertheless has a + and – charge in it.
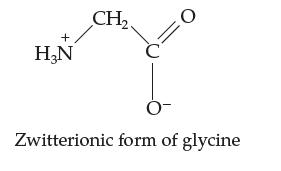
Now, the NH3+ end is weakly acidic and the COO– end is weakly basic. Does this mean that the zwitterionic form cannot supply buffering action against strong acid or strong base in water? Explain
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:





