Balance each equation and assign each a reaction type: (a) SiO(s) + C(s) Si(s) + CO(g) H
Question:
Balance each equation and assign each a reaction type:
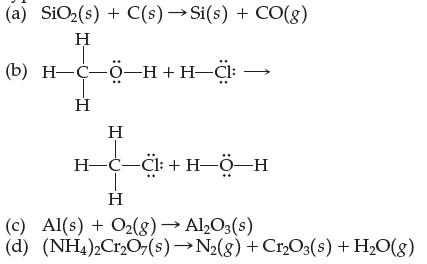
Transcribed Image Text:
(a) SiO₂(s) + C(s) →Si(s) + CO(g) H (b) H-C-Ö-H+H-Cl: H H H-C-C1:+H-Ö-H T H (c) Al(s) + O₂(g) → Al2O3(s) (d) (NH4)2Cr₂O7(s)→N₂(g) + Cr₂O3(s) + H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a SiO2 Cs Si CO2 b CH OH HCl ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Balance this chemical equation and assign it a reaction type: SO 2 (g) + O 2 (g) SO 3 (g)
-
1A) Assign an oxidation number to each atom in thereactants. Na2S(aq)+NiCl2(aq)?2NaCl(aq)+NiS(s) 1B) Assign an oxidation number to each atom in theproducts. Na2S(aq)+NiCl2(aq)?2NaCl(aq)+NiS(s) 1C) Is...
-
The chemical equation for the reaction of baking soda (sodium bicarbonate, NaHCO 3 ) and vinegar (acetic acid, CH 3 COOH) may be written as two steps. Fill in the missing information for the chemical...
-
Use implicit differentiation to find dy/dx. 6x 3 + 7y 3 = 13xy
-
How would the algebraic formula used to compute the break-even point under the equation method be changed to solve for a desired target profit?
-
Marino Company predicts that it will use 25,000 units of material during the year. The expected daily usage is 200 units, and there is an expected lead time of five days and a desired safety stock of...
-
Why can damage to one side of the cerebrum affect the functioning of the opposite side of the body?
-
On April 1, 2014, Maria Adams established Custom Realty. Maria completed the following transactions during the month of April: Amy Austin established an insurance agency on March 1 of the current...
-
The three objects in the figure are connected by a light cord.A triangular structure is oriented such that its base rests upon a horizontal surface, its right side is perpendicular to its base, and...
-
Aqueous solutions of sodium sulfide and iron(III) nitrate are combined. (a) Does a precipitation reaction occur? Explain. (b) If it does, write a net ionic equation for the reaction.
-
You need to prepare some calcium nitrate. (a) Write an intact-molecule balanced equation for an acidbase neutralization reaction that could be used to prepare calcium nitrate. (b) Write the net ionic...
-
In what ways has the scope of marketing research in healthcare broadened, and what accounts for the expanded scope?
-
Draw block diagram showing the main components of a computer.
-
What is stand-alone risk?
-
What is the inpatient prospective payment system (IPPS), and how does it work?
-
What is cost shifting?
-
What reimbursement methodology is unique to managed care plans?
-
In 2007, Charles Riegel and his wife sued a medical device manufacturer, Medtronic. Charles had a catheter produced by Medtronic placed in his coronary artery after he suffered a heart attack....
-
(a) Given a mean free path = 0.4 nm and a mean speed vav = 1.17 105 m/s for the current flow in copper at a temperature of 300 K, calculate the classical value for the resistivity of copper. (b)...
-
An optical filter can be described by a Jones matrix Obtain the form of the emerging light for each of the following incident beams: (a) A plane polarized beam polarized at angle θ to...
-
Keeping Eq. (8.33) in mind, write an expression for the randomly polarized flux density component (I n ) of a partially polarized beam in terms of the Stokes parameters. To check your result, add a...
-
Construct a Mueller matrix for an isotropic plate of absorbing material having an amplitude transmission coefficient of t. What Mueller matrix will completely depolarize any wave without affecting...
-
Ivanhoe Inc.'s books revealed the following data at year end after all adjustments were made: Cash sales $766000 Sales returns (on credit sales) 31000 Allowance for expected credit losses (credit...
-
What laws does military drones violate in war and law enforcement? List the laws and argue for and against each.
-
Judges at times are not given a lot of lee way in their decision making when it comes to sentencing. Do you think the judicial system needs to afford them more discretion?

Study smarter with the SolutionInn App


