Circle the correct choice to indicate how many electrons each element must gain or lose to form
Question:
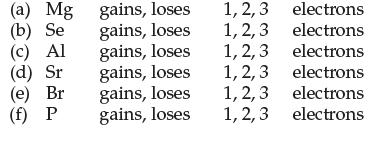
Circle the correct choice to indicate how many electrons each element must gain or lose to form an octet:
Transcribed Image Text:
(a) Mg (b) Se (c) Al (d) Sr (e) Br (f) P gains, loses gains, loses gains, loses gains, loses gains, loses gains, loses 1, 2, 3 1, 2, 3 1, 2, 3 1, 2, 3 1, 2, 3 1, 2, 3 electrons electrons electrons electrons electrons electrons
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Answer a Mg lose 2 electrone to from an octet because The electronic configuration of Mg is 1s 2 2s ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
1 points Question 11 Your 91-year-old great-aunt has terminal cancer. You and shehave had many conversations regarding what she would want done ifshe were incapacitated and could not make choices...
-
Marblehead Manufacturing, Inc., has two departments, Mixing and Blending. When goods are completed in Mixing, they are transferred to Blending and then to the finished goods storeroom. There was no...
-
Winchester Corporation makes printed cloth in two departments: weaving and printing. Currently, all product first moves through the weaving department and then through the printing department before...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Woolford Inc. declared a cash dividend of $1.00 per share on its 2 million outstanding shares. The dividend was declared on August 1, payable on September 9 to all stockholders of record on August...
-
3. 1. Find the volume of the figures below. 2. Answer: Answer: 8.5 ft 4 ft. 2.5 ft. 7 cm. 14 cm. 6 cm. 4. 12 in 4 in. Answer: 9 in. Juice Answer: 6.5 in. 3 in. 2.5 in. 5 in.. 3 in.
-
What are the group number, period number, and name of the element whose electron configuration is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10 4p 2 ?
-
If gamma radiation has a wavelength of 1.00 10 12 m, what is the energy of gamma radiation in joules?
-
Mary paid $2,000 of state income taxes in 2012. The total sales tax she paid during 2012 was $5,500, which included $3,000 for the cost of a new car. How should Mary treat the taxes paid on her 2012...
-
On January 15, 2020, the U.S. Treasury issued a 10-year inflation-indexed note with a coupon of 7%. On the date of issue, the CPI was 412. By January 15, 2030, the CPI had decreased to 279. What...
-
Suppose Microsoft has no debt and a WACC of 9.2%. The average debt-to-value ratio for the software industry is 8.6%. What would be its cost of equity if it took on the average amount of debt for its...
-
HighGrowth Company has a stock price of $18. The firm will pay a dividend next year of $1.12, and its dividend is expected to grow at a rate of 3.6% per year thereafter. What is your estimate of...
-
You are finalizing a bank loan for $205,000 for your small business and the closing fees payable to the bank are 1.7% of the loan. After paying the fees, what will be the net amount of funds from the...
-
Book Co. has 1.8 million shares of common equity with a par (book) value of $1.05, retained earnings of $28.3 million, and its shares have a market value of $48.93 per share. It also has debt with a...
-
Williams Manufacturing uses scrap metal to produce various tools, such as drill bits, hammer heads, saw blades, and nails. The CEO has asked you to analyze the saw blades division to determine asset...
-
What are some of the features of the Unified Process (UP)?
-
In Figure 19.16, n J /n 0 increases initially with J for all three temperatures for CO, but only for the two highest temperatures for HD. Explain this difference. Figure 19.16 HD CO 2.5 12 10 2.0...
-
What is the difference between the transition dipole moment and the dynamic dipole moment?
-
Nitrogen and oxygen do not absorb infrared radiation and are therefore not greenhouse gases. Why is this the case?
-
Analyze how social practices have been shaped by cultural diversity in modern culture.
-
"What is the influence of social comparison processes, reference group dynamics, and perceived fairness perceptions on motivation levels, effort allocation, and performance outcomes within...
-
Explain how integrating the four lenses helps people to understand how problems in diversity impact others in both professional and personal contexts.

Study smarter with the SolutionInn App


