Consider the following reaction. Use labeled arrows to indicate which bonds must be broken and which bonds
Question:
Consider the following reaction. Use labeled arrows to indicate which bonds must be broken and which bonds must be formed.
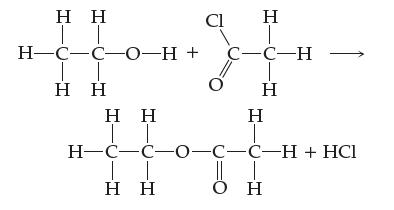
Transcribed Image Text:
Η Η ΤΤ H-CC-0-H + Η Η Η Cl Η Τ H¬C-C-0-C-C-H + HCl Η Η H C-C-H Η Η Ο Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Bonds broken The CO bond in the starting material HCCOH The HH bond in the hydrogen chlori...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Imagine a collision between an oxygen, O 2 , molecule and a hydrogen, H 2 , molecule to give the product hydrogen peroxide, H 2 O 2 . Draw Lewis dot diagrams for all the molecules, and then indicate...
-
Describe the following reaction in terms of which bonds must be broken and which bonds must be formed: HC=CH + 30 2 COz+2 H,O
-
Is it likely that a single collision leads to the breaking of all the bonds that you named in Problem 13.27? Explain your answer. Data from Problem 13.27 Describe the following reaction in terms of...
-
Draw an ER model according to the below information. NOTE: Use a word or any website to produce the diagrams. Dont use handwriting. In ERD diagram: 1. Entity: There are 6 Entity in this diagram:...
-
Venus Chocolate Company processes chocolate into candy bars. The process begins by placing direct materials (raw chocolate, milk, and sugar) into the Blending Department. All materials are placed...
-
Presented below is financial information for two different companies.Instructions(a) Determine the missing amounts.(b) Determine the gross profit rates. (Round to one decimalplace) Dae Company Kim...
-
In Figure P8.35, a \(50-\mathrm{kg}\) skier heads down a slope, reaching a speed of \(35 \mathrm{~km} / \mathrm{h}\). She then slides across a horizontal snow field but hits a rough area. Assume the...
-
David, Matt, and Chris are forming The Doctor Partnership. David is transferring $30,000 of personal cash and equipment worth $25,000 to the partnership. Matt owns land worth $18,000 and a small...
-
a) Solve cos x = 2xy and cos xy = 2x to 5 decimal places with an initial guess of x0 = 0.5 and yo= 0.5 using proper method. (90 Point) ATTENTION: Please add a comment line to each line of code...
-
What is meant by the mechanism of a chemical reaction?
-
Indicate if the following reactions are examples of a substitution reaction. Explain for each. (a) CHI+ Cl CH3Cl + I (b) NH3 + CHI[(CH3)NH]*I
-
The probability that exactly eight defective parts are in the shipment A discrete random variable is given. Assume the probability of the random variable will be approximated using the normal...
-
Back in the 1990s, drug kingpin Pablo Escobar couldn't use a bank account for all his cash so he stored paper US dollar bills in a warehouse. About 10% of the money was lost because of water damage,...
-
How do cultural differences impact leadership styles and practices, and what strategies can leaders employ to effectively lead diverse teams and navigate cross-cultural challenges ?
-
How has the pharmaceutical industry had a history of failing to foster inclusion in the decision-making process? Provide specific examples. How does practicing inclusion (recognizing that...
-
How do you currently or how will you be your student's champion? The introduction of The Listening Leader lists what a transformational school looks like (xxx). Do you agree or disagree with this...
-
2. Graph y(x) = x. Express the domain and range for this function in interval notation.
-
Troy's 2009 tax return is audited. The auditor determines that Troy inadvertently understated his ending inventory in calculating his business income. The error creates an additional tax liability of...
-
Which task is performed by a book-keeper? A. Analysing the trading results B. Entering transactions in the ledger C. Preparing year-end financial statements D. Providing information for...
-
A convex-planar lens of index 3/2 has a thickness of 1.2 cm and a radius of curvature of 2.5 cm. Determine the system matrix when light is incident on the curved surface.
-
A thick biconvex lens in air has an index of 1.810 and a thickness of 3.00 cm. Its first radius of curvature is 11.0 cm and its second is 120 cm. Determine its system matrix A.
-
Starting with Eq. (6.33) derive Eq. (6.34) when both the object and image are in air. s-ein e | 1 1 @ a12 1||a21 a22]-do/no o lu/'p (6.33) olaz1 az2do + (j1 12dojdj] (6.34) + yolaz2 + a12d)
-
Zoukis Forklift Services Ltd General Ledger Year end December 3 1 st 2 0 2 3 Date Details Folio # Dr Cr 0 1 - Jan Cash 1 1 $ 5 0 0 , 0 0 0 Accounts Receivable 1 2 $ 1 5 0 , 0 0 0 Supplies 1 4 $...
-
Explain to what extent you believe that a person's psychology has a direct impact on how they live out their faith. Where, if at all, is there a connection between one's psychology and spirituality?...
-
Calculate the molar mass of a compound in the Dumas method at 100C for which the volume of the experimental container was 452 ml and the pressure was 745.1 torr. The difference in mass between the...

Study smarter with the SolutionInn App


