For those reactions in Problem 10.58 that are electron-transfer reactions: (a) Indicate which atoms get oxidized and
Question:
For those reactions in Problem 10.58 that are electron-transfer reactions:
(a) Indicate which atoms get oxidized and which atoms get reduced.
(b) Indicate which reactant is the oxidizing agent.
(c) Indicate which reactant is the reducing agent.
Data from Problem 10.58
Which of the following are electron-transfer reactions?
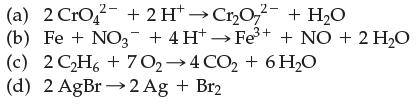
Transcribed Image Text:
(a) 2 CrO2 + 2H+→Cr₂O72- + H₂O (b) Fe + NO3 + 4H+→Fe³+ + NO + 2 H₂O (c) 2C2H6+7O,→4CO,+6H,O (d) 2AgBr→2Ag + Br2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
For those reactions in Problem 10.56 that are redox reactions: (a) Indicate which atoms get oxidized and which atoms get reduced. (b) Indicate which reactant is the oxidizing agent. (c) Indicate...
-
From the Nerst equation E = E0 + RT/nF ln a... show that for a pH electrode that an uncertainty in the measurement of the potential leads to an error of 4% in the measurement of the pH.
-
The following half reactions play important roles in metabolism. 1/2 O2 1 2H+ + 2e H2O NADH + H+ NAD+ + 2H+ + 2e- Which of these two is a half reaction of oxidation? Which one is a half reaction of...
-
The total cost of 2 chair and 1 table is 210 dollars. The total cost of 1 chair and 2 tables is 285 dollars. What is the cost of 1 chair?
-
The controller of Sedona Housewares Inc. instructs you to prepare a monthly cash budget for the next three months. You are presented with the following budget information: The company expects to sell...
-
The trial balance of the Kellen Davis Company shown below does not balance. Your review of the ledger reveals that each account has a normal balance. You also discover the following errors. 1. The...
-
Why does a dropped dinner plate break when it lands on a tile floor but not when it lands on carpet?
-
Three departmentsmilling (M), drilling (D), and sawing (S)are assigned to three work areas in Samuel Smiths machine shop in Baltimore. The number of work pieces moved per day and the distances...
-
Consider FED's problem that is described in the previous problem. To meet with state audit requirement, FED now has to ensure that it does not hold eggs for more than 20 days. 1. Does the current...
-
The following reaction is responsible for producing electricity in your car battery (often called a lead storage battery): (a) Assign an oxidation state to each atom. (b) Identify the atom that gets...
-
Which of the following are redox reactions? (a) 2Na + 2H 2 O 2NaOH + H 2 (b) MgBr 2 + 2NaF MgF 2 + 2NaBr (c) 2CO + O 2 2CO 2 (d) SO 2 + H 2 O H 2 SO 3
-
Sketch the streamlines and potential lines of the flow due to a line source of at (a, 0) plus an equivalent sink at (a, 0).
-
Two all around enhanced portfolios An and B (all quirky danger has been differentiated away) have 1) expeccted returns of 12% and 9% separately. The Beta for Portfolio An is 1.2 while the Beta for...
-
A sample of gas occupies a volume of 260 ml when the temperature is 85C and the pressure is 750mm Hg. Both the temperature and pressure were changed causing the volume occupied by the gas to become...
-
3. (20 points) Large-scale pathloss a. (12 points) Given are the following system design characteristics. Transmit power = 2W Frequency = 2.4 GHz Pathloss exponent = 3.5 Isotropic receiver antenna...
-
Before and after-tax cost of debt For the following $1,000-par-value bond paying semi-annual interest payments, calculate the before- and after-tax cost of debt. Use the 21% corporate tax rate....
-
Give an exar example to show that the result of the preceding problem need not hold when k is even.
-
Based solely on the definitions in the chapter, is the sales tax a proportional, regressive, or progressive tax? Explain, and state how the tax might be viewed differently.
-
In a certain school district, 3% of the faculty use none of their sick days in a school year. Find the probability that 5 faculty members selected at random used no sick days in a given year.
-
A candle flame is 18.0 cm in front of a thin positive lens. Its image appears three times farther away from the lens than if the same candle were on a very distant mountain. Determine the lenss focal...
-
What must the focal length of a thin negative lens be for it to form a virtual image 50 cm away (measured from the lens) of an ant located 100 cm away (measured from the lens)? Given (just as a...
-
An LED is on the central axis 30.0 cm in front of a thin lens. The resulting image, which is virtual, is 10.0 cm from the lens. Determine the focal length of the lens. Using Table 5.3, explain why...
-
Calculate the NPV of the Project using a discount rate of 15%. Year 0 = -$100; Year 1 = $50; Year 2 = $40; Year 3 = $40; Year 4= $15.
-
In class, you have learned that the break-even price for a call option is X + premium. Given that most of Finance formula does not drop out from the sky (i.e. there is a reason why the formula looks...
-
One method that works well for disseminating major changes in my current company are video conferences we call WebEx's. Something similar to this type of communication would be beneficial in this...

Study smarter with the SolutionInn App


