How many valence electrons does each atom in the methane molecule have? Double-count shared electrons. HTCIH
Question:
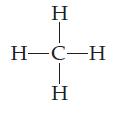
How many valence electrons does each atom in the methane molecule have? Double-count shared electrons.
Transcribed Image Text:
HTCIH Η H-C-H Η
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
The carbon atom has eight val...View the full answer

Answered By

Shebla K
I am an MBA graduate having experience as an Assistant Professor at University level for two years. I always prepare well for a class as I believe that only if you become an ocean you can give a bucket of water. Being a teacher was not only my profession but also my passion.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Explain subsequent event and why these are important? Explain with the help of example
-
How many valence electrons does a carbon atom normally have? How many valence electrons does it have in the methane molecule? How many electrons did it gain, and how many covalent bonds to hydrogen...
-
How many valence electrons does each of the following atoms have? (a) Na (b) Cl (c) Si (d) B (e) Ne (f) N
-
"One need not be concerned with reliability and validity in applied marketing research." Discuss this statement as a small group.
-
Accenture is a firm that provides a wide range of consulting and services to organizations worldwide. With more than 170,000 employees, the firm has clients in 120 countries that receive many HR and...
-
On January 1, 20X1, partners Art, Bru, and Chou, who share profits and losses in the ratio of 5:3:2, respectively, decide to liquidate their partnership. The partnership trial balance at this date...
-
Plaintiffs W. O. and J. C. Lucy had wanted to purchase Ferguson Farm from the Zehmers for at least eight years. One night, Lucy stopped by the establishment the Zehmers operated and said that he bet...
-
Rachel Rey recently opened her own basketweaving studio. She sells finished baskets in addition to the raw materials needed by customers to weave baskets of their own. Rachel has put together a...
-
A contractor has to move 15 300 Bank m 3 of wet sandy material in rear dump trucks, which will be loaded by an excavator. Average face depth will be 2.4 m with 60-90 degree average swing angle. Ten...
-
A covalent bond: (a) Is the name given to the attractive force between atoms in a molecule. (b) Is strong because the electrons in the bond can be attracted to two nuclei instead of just one. (c) Is...
-
Predict the formula of the compound that forms between silicon (Si) and bromine (Br).
-
The molecular formula of n-decane is CH 3 (CH 2 ) 8 CH 3 . Decane is not considered a polymer, whereas polyethylene is. What is the distinction?
-
The following table lists the tax rates for the United Kingdom in 2018: For income levels of $10,000; $30,000; $50,000; $140,000; and $200,000, calculate taxes due and compute the average tax rate....
-
A sporting goods store has estimated the demand curve for a popular brand of running shoes as a function of price. Use the diagram to answer the questions that follow. a. Calculate demand elasticity...
-
Kristine is a fan of both action movies and classical concerts. This month she has 480 to spend on the two activities. The original budget constraint is shown in the graph below. Let X represent...
-
Carmen has $84 to spend on California rolls and eel sashimi, and the data in the following table represents an indifference curve for these two products. If California rolls are $4.00 each and eel...
-
The following table shows the average annual growth rate in real GDP per capita for Argentina, Ghana, and South Korea using data from the World Bank, World Development Indicators, for the past few...
-
List important measures in assessing marketing effectiveness.
-
Which of the following streaming TV devices does not involve use of a remote controller? A) Google Chromecast B) Apple TV C) Amazon Fire TV D) Roku
-
Consider rotation about the C~C bond in ethane. A crude model for torsion about this bond is the free rotor model where rotation is considered unhindered. In this model the energy levels along the...
-
Inspection of the thermodynamic tables in the back of the text reveals that many molecules have quite similar constant volume heat capacities. a. The value of C V, m for Ar (g) at standard...
-
The molar constant volume heat capacity for I 2 (g) is 28.6 J mol 1 K 1 . What is the vibrational contribution to the heat capacity? You can assume that the contribution from the electronic degrees...
-
The demand for Oman Food company is 45000 units daily. In the past they had faced a lot of issues related with ordering and carrying cost. Mr. Mohammed has joined this company recently and gave a...
-
why would you need these discussions with management in concluding on the severity of a deficiency? (hint: who is ultimately responsible for the company's control environment, the auditor, or...
-
You discuss a couple of the learning groups here. When you took the quiz, what were your results? Were you a visual learner? I agree there are people that benefit from more than one learning style. I...

Study smarter with the SolutionInn App


