Suppose you have 120.11 g of carbon atoms. (a) How many moles of carbon atoms do you
Question:
Suppose you have 120.11 g of carbon atoms.
(a) How many moles of carbon atoms do you have?
(b) How many carbon atoms do you have?
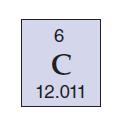
Transcribed Image Text:
6 C 12.011
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a To find the number of moles of carbon atoms Number of moles Mass Mo...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Mole Lab will rate answer Mole Laboratory Materials & Apparatus: Scale Graduated cylinder(s) Paper or plastic cups (8 oz or 16 oz) New container of small candy (jelly beans, Skittles, M&Ms,or...
-
The following are the financial statements of Swifty Corporation. Swifty Corporation Comparative Balance Sheets December 31 Assets 2019 2018 Cash $37,200 $19,700 Accounts receivable 33,000 18,400...
-
If you have 1 mole of glucose (C 6 H 12 O 6 ): (a) How many moles of carbon atoms do you have? (b) How many moles of hydrogen atoms do you have? (c) How many oxygen atoms do you have? (Note that we...
-
Evaluate the limit or determine that it does not exist. |xx| lim (x,y)(0,0) [x] + [yl
-
A local government agency has asked you to consult regarding acquisition of land for recreation needs for the urban area. The following data are provided: Urban population 10 years ago 49,050 Urban...
-
Review the methods for dealing with resistance to change. Generate specific examples of each that you have seen, and analyze the reasons why they worked or failed to work.
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
On December 31, 2016, Akron, Inc. purchased 5 Percent of Zip Company's common shares on the open market in exchange for $16,000. On December 31, 2017, Akron, Inc., acquires an additional 25 percent...
-
John just sold the home where he's lived for eight years and made a nice profit. Which of these statements about the profit he made is FALSE? Unset starred question He can include depreciation costs...
-
In grams, one mole of a molecule weighs (a) The sum of its constituent element atomic masses, each multiplied by its subscript in the formula. (b) Its molar mass in grams. (c) 6.022 10 23 times the...
-
One mole of an element weighs (a) Its atomic mass divided by 6.022 10 23 /mol. (b) Its atomic mass times 6.022 10 23 /mol. (c) Its atomic mass listed on the periodic table in grams. (d) One-twelfth...
-
The active ingredient in aspirin is acetylsalicylic acid. A 2.51-g sample of acetylsalicylic acid required 27.36 mL of 0.5106 M NaOH for complete reaction. Addition of 15.44 mL of 0.4524 M HCl to the...
-
Two blocks of masses m = 13,7 kg and m = 9, 4 kg are connected by a rope that hangs over a pulley as shown in the figure. The pulley is a uniform disk of radius R = 0,5 m and the mass M = 8,9 kg. The...
-
Examine the following table of values of a quadratic function f. X -5 -4 -3 -2 -1 0 f(x) -6 -3 -2 -3 -6 -11 a. What is the equation of the axis of symmetry of the associated parabola? Justify your...
-
The current in a 150 resistor is i = 10e-2tu(t) A. What is the energy dissipated in the resistor associated with the frequency band 0 w 23 rad/s? Hint1. Parseval's theorem states that the energy of...
-
The function Y is defined as Y + k. Based on the table shown, what is the value of k? k XO-2345 VROLONG 0 1 Y 15 10 7 6 7 10 Y VAWANNN 12
-
If f(x) = 6 + Find '(1). Find (x). Find f"(1). 7 X + 2 2 find f'(x). > O
-
The U.S. Department of Agriculture publishes data annually on various selected farm products. Shown here are the unit production figures for three farm products for 10 years during a 20-year period....
-
Highland Theatre is owned by Finnean Ferguson. At June 30, 2014, the ledger showed the following: Cash, $6,000; Land, $100,000; Buildings, $80,000; Equipment, $25,000; Accounts Payable, $5,000;...
-
The two shafts are made of A-36 steel. Each has a diameter of 25 mm and they are connected using the gears fixed to their ends. Their other ends are attached to fixed supports at A and B. They are...
-
The two shafts are made of A-36 steel. Each has a diameter of 25 mm and they are connected using the gears fixed to their ends. Their other ends are attached to fixed supports at A and B. They are...
-
A rod is made from two segments: AB is steel and BC is brass. It is fixed at its ends and subjected to a torque of T = 680 N m. If the steel portion has a diameter of 30 mm, determine the required...
-
How to convert this Entity-Relationship data model into a relational database model? Contains Inventory Inventory ID Customer Cust_ID Amount Availible Cust LName Must Have Coffee_Type Cust Fname...
-
What other options exist if you do not want to use the relational model? Researchon the Internet and find one other type of database model, describe that model, and post the link to the source that...
-
What do you mean by Hardware Software Interface? Give a concrete example. Define the two principle integrity rules for the relational model. Discuss why it is desirable to enforce these rules.

Study smarter with the SolutionInn App


