Taking all configurations to be for neutral atoms, identify the elements having the following electron configurations: (a)
Question:
Taking all configurations to be for neutral atoms, identify the elements having the following electron configurations:
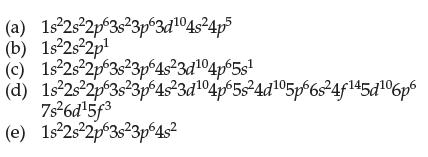
Transcribed Image Text:
(a) 1s²2s²2p63s 3p63d¹04s²4p5 (b) 1s²2s²2p¹ (c) 1s²2s22p63s23p64s²3d¹04p65s¹ 1s²2s22p63s23p64s²3d¹04p65s²4d¹05p 6s²4f¹45d¹06p6 (d) (e) 7s²6d¹5f3 1s²2s²2p63s²3p64s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Lets identify the elements corresponding to the given electron ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
You are working as a project manager and have identified standards and quality requirements for both the product and project. Which stage of quality management are you in? Select one: Quality...
-
The electron configurations described in this chapter all refer to gaseous atoms in their ground states. An atom may absorb a quantum of energy and promote one of its electrons to a higher-energy...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
1. Rick and Barbara are married with two children. Four yearsago, Barbara bought a $75,000 life insurance policy on her motherslife and named her children as policy beneficiaries. Barbara didnot name...
-
Folsom Fabricators, Inc., uses a standard cost system to account for its single product. The standards established for the product include the following: Materials . . . . . . . . . . . . . . . . . ....
-
Gormley Precision Tools makes cutting tools for metalworking operations. It makes two types of tools: A6, a regular cutting tool, and EX4, a high-precision cutting tool. A6 is manufactured on a...
-
What is the internal rate of return of the following cash flow diagram? a. 20 percent b. 18.2 percent c. 17.5 percent d. 15 percent $30 $31 0 1 2 3 $30 $15
-
Colaw Stores accepts both its own and national credit cards. During the year, the following selected summary transactions occurred. Jan. 15 Made Colaw credit card sales totaling $18,000. (There were...
-
Two identical loudspeakers are driven in phase by a common oscillator at 880 Hz and face each other at a distance of 1.22 m. Locate the points along the line joining the two speakers where relative...
-
Draw a simple Bohr model (no subshells) for an oxygen atom. How many electrons are in the valence shell? How many more electrons can be put into the valence shell?
-
Rank visible light, gamma rays, X-rays, radio/television waves, infrared radiation, and ultraviolet light in order of (a) Increasing wavelength (b) Increasing energy.
-
Suppose the income statement for Goggle Company reports $70 of net income, after deducting depreciation of $35. The company bought equipment costing $60 and obtained a long-term bank loan for $60....
-
Gamlouch Industries uses flexible budgets to control its selling expenses. Monthly sales are expected to be from $200,000 to $240,000. Variable costs and their percentage relationships to sales are:...
-
The amount of fair value changes taken to equity as at 31 December 20x5 was: (a) $10,000 (b) $20,000 (c) $60,000 (d) $70,000 (e) None of the above Alpha Corporation had the following transactions in...
-
Eastern Company had inventory of 5,000,000 one-ounce silver coins, which were carried at a cost of $15 million (current market value is $16.5 million). The selling price of the silver coins was...
-
On 1 January 20x1, Delco Company initiated a share-based compensation plan for its ten employees. Under the plan, each employee was given two choices: either a right to a cash payment equal to the...
-
On 31 December 20x0, Carmen Corporation granted 10,000 share options to 35 employees. The share options would vest at 31 December 20x3 provided the employees remained in service until then. The share...
-
Felton Co. produces rubber bands for commercial and home use. Felton reported $1 million residual income (RI) with $20 million net book value (NBV) of assets and $5 million in income for the year....
-
In your readings, there were many examples given for nurturing close family relationships in this ever-evolving technological society we live in Based upon your readings and research describe three...
-
A coin with a mass of 8.31g suspended on a rubber band has a vibrational frequency of 7.50 s 1 . Calculate a. The force constant of the rubber band b. The zero point energy, c. The total vibrational...
-
Show that the function Y 2 0 (, ) = (5/16) 1/2 (3cos 2 1) is normalized over the interval 0 and 0 2.
-
Is it possible to simultaneously know the angular orientation of a molecule rotating in a two-dimensional space and its angular momentum? Answer this question by evaluating the commutator [, ih(/]
-
In two-slit interference, if the slit separation is 17 m and the slit widths are each 2.9 m, (a) how many two-slit maxima are in the central peak of the diffraction envelope and (b) how many are in...
-
Your division is considering two projects. Its WACC is 10%, and the projects' after-tax cash flows (in millions of dollars) would be as follows: Expected Cash Flows Time Project A Project B 0 ($30)...
-
A rock thrown with speed 1 2 . 0 m / s and launch angle 3 0 . 0 ( above the horizontal ) travels a horizontal distance of d = 1 7 . 5 m before hitting the ground. From what height was the rock...

Study smarter with the SolutionInn App


