The boiling point of one of these compounds is 24 C, and the boiling point of the
Question:
The boiling point of one of these compounds is –24 °C, and the boiling point of the other is 78°C:
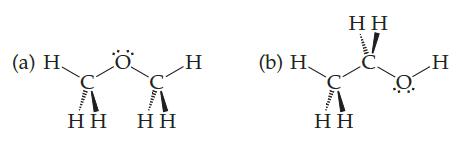
Which boils at which temperature? Why? If hydrogen bonds are involved in either case, make a drawing using dotted lines to show the hydrogen bonding
Transcribed Image Text:
(a) H Η Η Ö Η Η H (b) Η, Η Η Η Η Ο H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Compound b boils at 78 C and compound a boils at24 C The reason cant be London forces beca...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The temperature at which water boils (the boiling point) depends on elevation: The higher the elevation, the lower the boiling point will be. At sea level, water boils at 212F; at an elevation of...
-
At sea level, water boils at a temperature of 212F. As the altitude increases, the boiling point of water decreases. For instance, at an altitude of 5000 feet, water boils at about 202.8F. (a) Find a...
-
Glorious Electrical Appliances (GEP) Co. is a company that sells electrical tools. GEP uses perpetual inventory system in recording its inventory. The financial position of GEP as at 31 December 2016...
-
H. S. Black is noted for developing a negative feedback amplifier in 1927. Often overlooked is the fact that three years earlier he had invented a circuit design technique known as feed forward...
-
Mass communications will play a key role in Sonics product introduction.After reviewing your earlier decisions and thinking about the current situation (especially your competitive circumstances),...
-
In 1972, Donald and Joyce Carnahan purchased a 1-acre lot located on a 22-acre lake. The purchase included a portion of the lake bed. The Carnahans used the lake for recreational activity in both...
-
1. Your instructor will divide the class into teams and assign each team the task of investigating the start-up of one of the following businesses: a. Submarine sandwich shop b. Day care service c....
-
Beverage Products Company specializes in 12-ounce drinking glasses. The president asks the controller to prepare a performance report for April. The following report was handed to her a few days...
-
distributed database management system, are there any concerns or issues managing distributed database management systems, and if so, why?
-
Draw a picture showing how NH 3 molecules attract one another.
-
The molecules in a gas phase will (a) Speed up upon heating the gas. (b) Slow down upon heating the gas. (c) Change phase upon sufficient heating of the gas. (d) Form a condensed phase upon...
-
The aldol reaction is catalyzed by acid as well as by base. What is the reactive nucleophile in the acid catalyzed aldol reaction? Propose a mechanism.
-
Green Energy Company files a suit against First Bank, claiming that the consideration for a contract between them was inadequate. The court likely will not evaluate the adequacy of consideration...
-
Lyydia works for Consolidated Manufacturing Company under a contract in which she agrees not to disclose any process she uses while in Consolidateds employ. When Lyydia goes into business for...
-
Damba offers to buy a book owned by Lee for $40. Lee accepts and hands the book to Damba. The transfer and delivery of the book constitutes performance. Is this performance consideration for Dambas...
-
Luke practices law without an attorneys license. The state requires a license to protect the public from unauthorized practitioners. Clark hires Luke to handle a legal matter. Luke cannot enforce...
-
Deb owes Jim $5,500. In need of the money, Jim threatens to foreclose on the debt. Deb offers to pay $5,000 immediately to settle Jims claim. Jim agrees. This is a. promissory estoppel. b. a release....
-
Steinberg Company had the following direct materials costs for the manufacturing of product T in March: Actual purchase price per pound ................... $7.50 Standard direct materials allowed for...
-
The area of square PQRS is 100 ft2, and A, B, C, and D are the midpoints of the sides. Find the area of square ABCD. B A
-
Identify the reagents necessary to make each of the following amino acids using a HellVolhardZelinski reaction. (a) Leucine (b) Alanine (c) Valine
-
Draw the aldehyde that is obtained as a byproduct when l-leucine is treated with ninhydrin.
-
Identify the starting alkene necessary to make each of the following amino acids using an asymmetric catalytic hydrogenation. (a) l-alanine (b) l-valine (c) l-leucine (d) l-tyrosine
-
Compare between Invasive and modern non-invasive prenatal tests.
-
This is a group discussion. Only members of your group (and the instructor) can see your posts here. Use this discussion to communicate and plan with your group members. This discussion is not...
-
Conflicts of interest generated due to different tax rates in MNC capital budgeting: Can be resolved using the currency content analysis technique can be resolved using the net present-value...

Study smarter with the SolutionInn App


