The radioactive isotope of iodine 123 53 I is used to treat thyroid disease. Suppose a patient
Question:
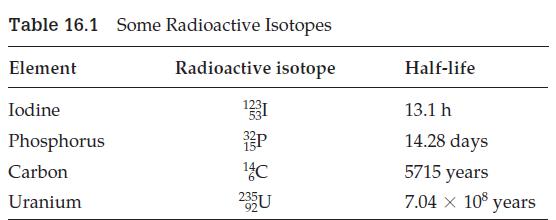
The radioactive isotope of iodine 12353I is used to treat thyroid disease. Suppose a patient is given a 30 mg dose. How much will be left in the patient after 39.3 h? (See Table 16.1.)
Transcribed Image Text:
Table 16.1 Some Radioactive Isotopes Radioactive isotope Element Iodine Phosphorus Carbon Uranium 1331 P ¹4 C 235U Half-life 13.1 h 14.28 days 5715 years 7.04 × 108 years
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
Because 393 h i...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
A breeder reactor converts stable uranium-238 into the isotope plutonium-239. The decay of this isotope is given by A(t) = A0e-0.00002876t where A(t) is the amount of the isotope at time t, in years,...
-
The radioactive isotope of iodine, 1- 1 35, decays into the radioactive isotope Xe- 1 35 of xenon; this in tum decays into another (stable) product. The half-lives of iodine and xenon are 6.7 hours...
-
Suppose a radioactive material has a half-life of 100 years. What is the decay rate ? Starting with an initial sample of 100 grams, how much will be left after 10 years? 100 years? 1,000 years?
-
Will restricting imports affect exports and export-related jobs? How?
-
A dialysis clinic provides two types of treatment for its patients. Hemodialysis (HD), an in house treatment, requires that patients visit the clinic three times each week for dialysis treatments....
-
Explain why opportunity costs are not included in the accounting records.
-
An electrolyte contains equal numbers of positive and negative ions in solution. When an electric field is applied, the positive ions move in the direction of the field and the negative ions move...
-
The Wall Street Journal routinely publishes summaries of corporate quarterly and annual earnings reports in a feature called the Earnings Digest. A typical digest report takes the following form. (a)...
-
3.1 Solve for x by completing the square: 6-4x= x 3.2 The length of a rectangle is three times as long as its width. The area of the rectangle is 90m. Calculate the length and the width of the...
-
The nuclear process which constantly produces 14 C in the Earths atmosphere is illustrated below, along with the mechanism of its incorporation into the food chain. What is the major assumption upon...
-
(a) What does the plot say about the stability of the nucleus of the iron isotope 56 26 Fe? (b) What is the total binding energy for the 56 26 Fe nucleus in kilojoules per mole of atoms?
-
Recall that the file Baseball Salaries Extra.xlsx contains data on 877 Major League Baseball (MLB) players during the 2018 season. Find the mean, median, standard deviation, and first and third...
-
Calculate the slope of the function you graphed in Problem A-6. Problem A-6 Given the function y = 8 - 2x, complete the following schedule and plot the curve. y X -4 -2 ON
-
A new product involves the following costs associated with three possible locations. If demand is forecast to be 3900 units a year, which location should be selected? A Annual cost ($) 10,000 Unit...
-
Briefly state the general principle that may be distilled from the post- Caparo cases reported in this chapter which the courts appear to seek to apply in cases involving auditors breach of their...
-
How do quality practices that originated in the manufacturing industry differ from the traditional quality practices of healthcare organizations?
-
The fermentation of cane sugar to ethanol was introduced in problem 4.3, and typical yields were given. Design the reactor system for a plant that produces 500,000 metric tons per year of ethanol...
-
What constitutes "reasonable accommodation" under the Rehabilitation Act and the ADA?
-
Is it ethical to provide safety training in English to immigrant workers who speak little English, in order to reduce costs?
-
The switch in Fig. 16.49 moves from position A to position B at t = 0 (please note that the switch must connect to point B before it breaks the connection at A, a make before break switch). Determine...
-
The switch in the circuit of Fig. 16.47 has been closed for a long time but is opened at t = 0. Determine i(t) for t > 0. i(t) 2 40 V t = 0 -/4 1/2
-
Find the voltage across the capacitor as a function of time for t > 0 for the circuit in Fig. 16.45 . Assume steady-state conditions exist at t = 0 . 5 t = 0 1 0.25 60 V (+ +1
-
Write a function named digit_count that takes one parameter that is a number (int or float) and returns a count of even digits, a count of odd digits, and a count of zeros that are to the left of the...
-
Can you elucidate the concept of taxonomic inflation and its implications for conservation prioritization and management, particularly in the context of recognizing and conserving evolutionary...
-
a) [5 marks] Why does the transport layer provide two major protocols, UDP and TCP? What is the purpose of each protocol? Describe the differences between TCP and UDP. b) [5 marks] Describe the...

Study smarter with the SolutionInn App


