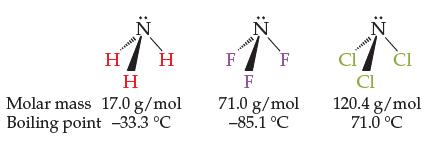
The three molecules below increase steadily in molar mass from left to right. However, their boiling points
Question:
The three molecules below increase steadily in molar mass from left to right. However, their boiling points do not. Explain why the boiling points do not also steadily increase from left to right.
Transcribed Image Text:
H H H Molar mass 17.0 g/mol Boiling point -33.3 °C F :2 F F 71.0 g/mol -85.1 °C CI CI CI 120.4 g/mol 71.0 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The trend in boiling points of the molecules N2 HF and Cl2 doesnt strictly follow the increase in mo...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Write a literature review for your study. See below for an example of a literature review. Your literature review should provide both analysis and synthesis of previous studies as related to the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Holly needs $21,800 worth of new equipment for his shop. He can borrow this money at a discount rate of 11% for a year. Find the amount of the loan Holly should ask for so that the proceeds are...
-
Discuss why making changes such as Mitsubishi did is important both legally and for improving HR management with the employees and managers.
-
Select the correct answer for each of the following questions. 1. According to ASC 270 and 740, income tax expense in an income statement for the first interim period of an enterprise's fiscal year...
-
Teddys daily budget constraint is shown in the following chart. Teddys employer pays him a base wage rate plus overtime if he works more than the standard hours. What is Teddys daily nonlabor income?...
-
A company processes a chemical, DX-1, through a pressure treatment operation. The complete process has two outputs, L and T. The January costs to process DX-1 are $50,000 for materials and $100,000...
-
A system consisting of a small 1.20-kg object attached to a Ax(cm) light spring oscillates on a smooth, horizontal surface. A 5.00- graph of the position x of the object as a function of time is 4.00...
-
What is wrong with the following diagram when it comes to explaining London forces?
-
Arrange in order of increasing boiling point: CO 2 , SO 2 , CH 3 CH 2 OH, Al.
-
Jansen Company reports the following for its ski department for the year 2017. All of its costs are direct, except as noted. Sales .................................... $605,000 Cost of goods sold...
-
What are the names of the following AASB standards? a AASB 101 b AASB 102 c AASB 107 d AASB 112 e AASB 116 f AASB 118 g AASB 119 h AASB 137 i AASB 138
-
For each of the business transactions listed below, you are to enter in the Workbook: a the account name, with the debit account first b whether the account entry is a debit or credit c the chart of...
-
Why do business transactions need to be documented?
-
Draw a graph of the supply and demand for the American dollar in the Australian market. Diagram the effect of each of the following on the exchange rate; state in words whether the effect is long,...
-
For each of the service business transactions of a medical doctor listed below, you are to enter in the Workbook: a the account name, with the debit account first b whether the account entry is a...
-
Refer to the journal entries made in 15-29. Provide an appropriate end-of-year closing entry for each of the following two independent situations: (a) The net overhead variance is closed entirely to...
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
You are a NASA engineer faced with the task of ensuring that the material on the hull of a spacecraft can withstand puncturing by space debris. The initial cabin air pressure in the craft of 1 atm...
-
Describe the random-walk model of diffusion. How is this model related to Brownian motion?
-
In the StokesEinstein equation that describes particle diffusion for a spherical particle, how does the diffusion coefficient depend on fluid viscosity and particle size?
-
Compare adverse impact and disparate treatment during the staffing process by providing specific examples in your response. Discuss the federal law or laws that may be violated when an adverse impact...
-
Can use the trap vector information 0x30 postToChat(R0) 0x31 player.getTilePos() --> R0, R1, R2 0x32 player.setTilePos(R0, R1, R2) 0x33 getBlock(R0, R1, R2) --> R3 0x34 setBlock(R0, R1, R2, R3) 0x35...
-
Introduce yourself as leader and post your Leader Within representation and description here.

Study smarter with the SolutionInn App


