Two different compounds, both consisting of sodium (Na) and oxygen (O), were analyzed. The data are given
Question:
Two different compounds, both consisting of sodium (Na) and oxygen (O), were analyzed. The data are given below:
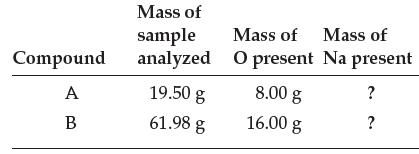
(a) Fill in the last column of the table.
(b) Calculate the %Na and %O for both compounds.
Transcribed Image Text:
Compound A B Mass of sample analyzed 19.50 g 61.98 g Mass of Mass of O present Na present ? ? 8.00 g 16.00 g
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
To fill in the last column of the table we need to calculate the mass of sodium Na prese...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The following questions concern Poisson regression models fit to fictitious follow-up study data in which rates of disease are modeled as a function of age and smoking status. The SAS program codes...
-
Which of the following is an example of how managers use production cost reports to control costs? A. providing cost of goods sold for the income statement B. determining if newer, more efficient...
-
In 1950, J. R. Clarkson founded a family-owned industrial valve design and manufacturing company in Sparks, Nevada. For almost a half century, the company, known as the Clarkson Company, worked on...
-
1. What options does Personal Trainer have for developing a new system? What are some specific issues and options that Susan should consider in making a decision? 2. Susan has been asked to prepare a...
-
The following information was summarized from the balance sheets of the Coca-Cola Company and Subsidiaries at December 31, 2008, and PepsiCo Inc. and Subsidiaries at December 27, 2008: Required 1....
-
Mazurka Corporation has 2,000 shares of $100, 8 percent cumulative preferred stock outstanding and 40,000 shares of $1 par value common stock outstanding. In the companys first three years of...
-
Continuing to focus on evidence associated with the act, concealment, and conversion, use the evidentiary material to continue the examination. In addition, as the examiner also start to think of...
-
English Motors, Ltd. (EML), developed a new all-wheel-drive sports utility vehicle. As part of the marketing campaign, EML produced a video tape sales presentation to send to both owners of current...
-
Discuss five Indirect or direct environmental issues of database management system. - discuss five Positive or negative economic effects of database management system.
-
Bromine (Br) has two abundant isotopes, one with 44 neutrons and the other with 46 neutrons. Give the full atomic symbols for both isotopes.
-
An electromagnet bends the path of the fast moving ions in a mass spectrometer so they can reach the detector. The strength of an electromagnets magnetic field depends on the how much voltage is...
-
Shareholders own the company. Why shouldnt they be entitled to any records they want?
-
What is a labor union?
-
What is a bond?
-
Katarina and Ivanna are sisters and plant lovers and have saved a total of $640 to spend on plants for their new apartment in Miami. They have decided to use this money on orchids and ferns. Their...
-
What is intra-industry trade?
-
What is command-and-control environmental regulation?
-
1 What are the effects of Johns time management for himself? What are the effects for the company? 2 Identify the tasks which John should have done himself and those which he should have delegated....
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Identify whether each of the following compounds exhibits a molecular dipole moment. For compounds that do, indicate the direction of the net molecular dipole moment: a. CHCl 3 b. CH 3 OCH 3 c. NH 3...
-
Which of the following compounds has the larger dipole moment? Explain your choice: CHCl 3 or CBrCl 3
-
The specific rotation of ephedrine in ethanol (at 20C) is -6.3. A chemist prepared a mixture of ephedrine and its enantiomer, and this mixture had a specific rotation of -6.0. Calculate the % ee of...
-
The employee credit union at State University is planning the allocation of funds for the coming year. The credit union makes four types of loans to its members. In addition, the credit union invests...
-
The stock price of Apax Incorporated is currently $105. The stock price a year from now will be either $130 or $90 with equal probabilities. The interest rate at which investors can borrow is 10%....
-
The current zero-coupon yield curve for risk-free bonds is as follows: Maturity (years) 5 5 YTM 4.96% 5.53% 5.78% 5.98% 6.04% What is the price per $100 face value of a four-year, zero-coupon,...

Study smarter with the SolutionInn App


