What is the volume in milliliters of 15.0 g of (a) Liquid water at 25C, (b) Ice
Question:
What is the volume in milliliters of 15.0 g of
(a) Liquid water at 25°C,
(b) Ice at 0°C,
(c) Gasoline at 25°C,
(d) Lead at 25°C,
(e) Mercury at 25°C,
(f) Helium gas at 0°C and 1 atm pressure?
(Use data from Table 2.4.)
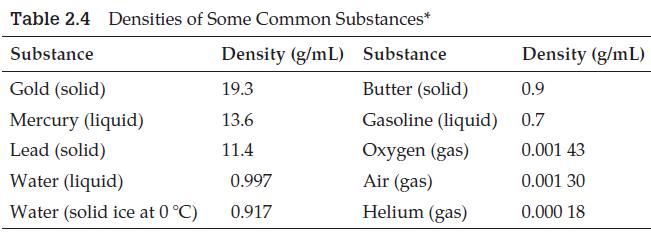
Transcribed Image Text:
Table 2.4 Densities of Some Common Substances* Substance Density (g/mL) Substance Gold (solid) 19.3 Butter (solid) Mercury (liquid) 13.6 Gasoline (liquid) Lead (solid) 11.4 Oxygen (gas) Water (liquid) Air (gas) Water (solid ice at 0 °C) Helium (gas) 0.997 0.917 Density (g/mL) 0.9 0.7 0.001 43 0.001 30 0.000 18
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a The density of liquid water at 25C is 0997 gmL so the volume of 150 g of liquid water a...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Determine x (1) 2 for the system of equations using GaussSeidel with an initial guess of x 1 = 0, x 2 = 0, x 3 = 0. 3 24 X1 [CHO 1 30 X2 24 1 X3 6 3 4
-
Liquid water at 25 C and 1 bar fills a rigid vessel. If heat is added to the water until its temperature reaches 50 C, what pressure is developed? The average value of ( between 25 and 50oC is 36.2 (...
-
A firm has total debt of $6,000,000 and stockholder's equity is $4,000,000. The firm wants to calculate equity-to- total asset ratio in order to make decision about further raise of capital. What is...
-
What are the advantages and disadvantages of team-written documents?
-
Yankee Athletic Club has preferred stock with a par value of $50 and an annual 6% cumulative dividend. Given the following market prices for the preferred stock, what is each investor seeking for his...
-
What must be included in a separate statement?
-
Abbott Corporation does not conduct a complete annual physical count of purchased parts and supplies in its principal warehouse but instead uses statistical sampling to estimate the year- end...
-
Mesas Plegadizas, S.A. fbrica mesas de diferentes tamaos y nos muestra su informacin del mes de noviembre: Madera $20,000 Sueldo supervisores $4,900 Bases metlicas $13,000 Sueldo cortadores $14,000...
-
The mass of an average neon atom is 20.2 atomic mass units (amu), where 1 amu = 1.66 10 24 g. (a) What is the mass in atomic mass units of 20 neon atoms? (b) What is the mass in grams of 20 neon...
-
A block measures 6.0 cm on each side. What is the volume of the block in cubic meters?
-
Find an equation for each graph. a. b. c. d. 1 -1 0 90 180 270 360
-
A 5.3 g coin sliding to the right at 23.0 cm/s makes an elastic head-on collision with a 15.9 g coin that is initially at rest. After the collision, the 5.3 g coin moves to the left at 11.5 cm/s. (a)...
-
Washington Brewery has two independent investment opportunities to purchase brewing equipment so the company can meet growing customer demand. The first option (equipment A) requires an initial...
-
(a) If A+B+C 180 prove that Sin(B+C-A)+Sin(C+A-B)+Sin(A+B-C)-Sin2A+Sin2B+Sin2C (b) Prove that Cos(x+y)Cos(x-y)-Cos'x-Sin'y-Cos'y-Sin'x
-
Calculate the average inventory (in $) and inventory turnover ratio for the given company. (Round the inventory turnover ratio to one decimal place.) Company Beginning Ending Average Inventory...
-
6) Find the acceleration in the following: a) A cheetah starts from rest and reaches a speed of 20 m/sec in 5 seconds b) A human from rest and reaches a speed of 10 m/sec in 5 seconds c) A car...
-
The number of megapixels in a digital camera is one of the most important factors in determining picture quality. But do digital cameras with more megapixels cost more? The following data show the...
-
The pendulum consists of two rods: AB is pin supported at A and swings only in the y-z plane, whereas a bearing at B allows the attached rod BD to spin about rod AB. At a given instant, the rods have...
-
Which of the following compounds below do you expect to have a longer λ max ?
-
Predict the expected λ max of the following compound:
-
When 5-deutero-5-methyl-1,3-cyclopentadiene is warmed to room temperature, it rapidly rearranges, giving an equilibrium mixture containing the original compound as well as two others. Propose a...
-
Use the line plot to answer the questions. Miles Run 8 888 x x 23. What is the difference between the longest distance run and the shortest distance run? 9 9 99 10 24. X If you add all the distances...
-
Chemet manufactures chemicals in a continuous process. The company combines various materials in a machine at the beginning of the process, and conversion costs are added evenly during the process....
-
What role does discourse play in the construction of social identities, and how does it influence the distribution of power in institutional settings ?

Study smarter with the SolutionInn App


