Which, if any, of these molecules are polar? For any molecule you classify as polar, show both
Question:
Which, if any, of these molecules are polar? For any molecule you classify as polar, show both the individual bond dipole moment vectors and the overall molecular dipole moment vector. Explain your answers.
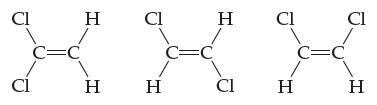
Transcribed Image Text:
Cl Cl C=C H H CI H C=C H CI Cl H C=C Cl H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Which if any of these molecules are polar The following molecules are polar Methanol CH3OH Water H2O Acetone C3H6O Ethanol C2H6O Individual bond dipol...View the full answer

Answered By

Saikumar Ramagiri
Financial accounting:- Journal and ledgers, preparation of trail balance and adjusted trail balance Preparation of income statement, retained earning statement and balance sheet Banks reconciliation statements Financial statement analysis Cash flow statement analysis (both direct and indirect methods) All methods of Depreciations Management Accounting:- Ratios Budgeting control Cash budget and production budget Working capital management Receivable management Costing:- Standard and variance costing Marginal costing and decision making Cost-volume-profit analysis Inventory management (LIFO, FIFO) Preparation and estimation of cost sheet Portfolio management:- Calculation of portfolio standard deviation or risk Calculation of portfolio expected returns CAPM, Beta Financial management:- Time value of money Capital budgeting Cost of capital Leverage analysis and capital structure policies Dividend policy Bond value calculations like YTM, current yield etc International finance:- Derivatives Futures and options Swaps and forwards Business problems Finance problems Education (mention all your degrees, year awarded, Institute/University, field(s) of major): Education Qualification Board/Institution/ University Month/Year of Passing % Secured OPTIONALS/ Major ICWAI(inter) ICWAI inter Pursuing Pursuing - M.com(Finance) Osmania University June 2007 65 Finance & Taxation M B A (Finance) Osmania University Dec 2004 66 Finance & Marketing. B.Com Osmania University June 2002 72 Income Tax, Cost & Mgt, Accountancy, Auditing. Intermediate (XII) Board of Intermediate May 1999 58 Mathematics, Accountancy, Economics. S S C (X) S S C Board. May 1997 74 Mathematics, Social Studies, Science. Tutoring experience: • 10 year experience in online trouble shooting problems related to finance/accountancy. • Since 6 Years working with solution inn as a tutor, I have solved thousands of questions, quick and accuracy Skills (optional): Technical Exposure: MS Office, SQL, Tally, Wings, Focus, Programming with C Financial : Portfolio/Financial Management, Ratio Analysis, Capital Budgeting Stock Valuation & Dividend Policy, Bond Valuations Individual Skills : Proactive Nature, Self Motivative, Clear thought process, Quick problem solving skills, flexible to complex situations. Achievements : 1. I have received an Award certificate from Local Area MLA for the cause of getting 100% marks in Accountancy during my Graduation. 2. I have received a GOLD MEDAL/Scholarship from Home Minister in my MBA for being the “Top Rank student “ of management institute. 3. I received numerous complements and extra pay from various students for trouble shooting their online problems. Other interests/Hobbies (optional): ? Web Surfing ? Sports ? Watching Comics, News channels ? Miniature Collection ? Exploring hidden facts ? Solving riddles and puzzles
4.80+
391+ Reviews
552+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Draw two ammonia molecules in their three-dimensional shape and show how they would be attracted to each other. Show the partial charges and individual bond dipole moment vectors for both molecules,...
-
Consider the molecule N 2 O (connected NNO). (a) Draw the dot diagram. (b) Draw the molecules three-dimensional shape, and label the numeric value of all bond angles. (c) What is the shape of this...
-
Consider the molecule SiCl 4 . (a) Draw the dot diagram. (b) Draw the molecules three-dimensional shape, and label the numeric value of all bond angles. (c) What is the shape of this molecule? (d)...
-
On December 1, 2016, Masipag sold land in exchange for a P180,000 non-interest, 1-year promissory note. The 10% interest rate was going market rate for similar notes. Masipag had paid P66,000 to...
-
Why are there objections to using absorption costing when segment reports of profitability are being prepared?
-
Given the income statement below for National Beverage Company for 2009, and the sales forecast from Problem 1, prepare a pro forma income statement for 2010. National Beverage Company Income...
-
Refer to information in QS 21-14. Compute the overhead volume variance for November and classify it as favorable or unfavorable. Data From QS 21-14 AirPro Corp. reports the following for November....
-
The comparative statements of Osborne Company are presented here. All sales were on account. Net cash provided by operating activities for 2014 was $220,000. Capital expenditures were $136,000, and...
-
1. Write an integral using cylindrical coordinates that represents the volume between the surfaces z=x+ y and z = 6-x - y.
-
Draw a combined Lewis dot, molecular-shape diagram for each of the following species. Name each shape, and indicate whether the molecule or ion has a dipole moment. If so, draw the dipole moment...
-
Draw a combined Lewis dot, molecular-shape diagram for each of the following species. Name each shape, and indicate whether the molecule or ion has an overall dipole moment. If so, draw the dipole...
-
Consider the mutually exclusive alternatives in Table P10-16. Which alternative would be chosen according to these decision criteria? a. Maximum benefit b. Minimum cost c. Maximum benefits minus...
-
Each of the following bond has contractual cash flows that meet the SPPI (solely payments of principal and interest) test. Determine the business model. (a) The entity invests surplus cash in short...
-
Describe, in your own words, the methodology of deferred tax accounting.
-
Go to the St. Louis Federal Reserve FRED database, and find data on potential output (GDPPOT), real GDP (GDPC1), a measure of the price level, the personal consumption expenditure price index...
-
How is a swap similar to a forward contract?
-
Explain the rationale for reporting diluted earnings per share.
-
(Multiple choice) 1. The primary objective of financial reporting is to provide information a. on the cash flows of the company. b. to the federal government. c. useful for making investment and...
-
White Bolder Investments (WBI) You are an intern working for WBI, a large investment advisory services in Sydney. Among other regular customers, WBI has been providing advisory services for Jumbo...
-
When 2-hepten-4-one is treated with LDA, a proton is removed from one of the gamma () positions. Identify which position is deprotonated, and explain why the proton is the most acidic proton in the...
-
When optically active (S)-2-methylcyclopentanone is treated with aqueous base, the compoundloses its optical activity. Explain this observation, and draw a mechanism that shows how racemization...
-
The racemization process described in the previous problem also occurs in acidic conditions. Draw a mechanism for the racemization process in aqueous acid.
-
Jenna bought a new car for $29,000. She paid a 10% down payment and financed the remaining balance for 48 months with an APR of 4.5%. Determine the monthly payment that Jenna pays.
-
Encik Aziz is the Head of Credit Division of CIMB Bank Berhad. Suggest any FOUR (4) strategies which Encik Aziz could adopt to manage credit risk in his bank. (16marks) Bank Negara Malaysia reduced...
-
Calculate the total interest expense on a $248,000 operating loan borrowed on February 1 at a 10% annual interest rate with repayments projected as $100,000 on August 1, $48,000 on September 1, and...

Study smarter with the SolutionInn App


