Which is the correct ground-state electron configuration for antimony, Sb? (a) 1s2s2p63s3p63d04s4p64d105s5d (b) 1s2s2b63s 3p63d04s4p64d05s5p3 (c) 1s2s2p63s3p64s3d04p65s24d05p4
Question:
Which is the correct ground-state electron configuration for antimony, Sb?
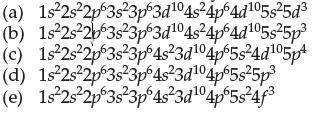
Transcribed Image Text:
(a) 1s²2s²2p63s²3p63d¹04s²4p64d105s²5d³ (b) 1s²2s²2b63s 3p63d¹04s²4p64d¹05s²5p3 (c) 1s²2s²2p63s²3p64s²3d¹04p65s24d¹05p4 (d) 1s²2s²2p63s²3p64s23d¹04p65s²5p3 (e) 1s 2s 2p 3s 3p 4s²3d¹04p5s²4f³
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Answer is b Atomic number of antimony ...View the full answer

Answered By

Anum Naz
Lecturer and researcher with 10+ years of experience teaching courses in both undergraduate and postgraduate levels. Supervised 17 BA theses, 07 MA theses, and 1 Ph.D. dissertations. Edited and co-authored 2 monographs on contemporary trends in political thought. Published over articles in peer-reviewed journals.
4.80+
11+ Reviews
51+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Specify the ground state electron configuration for silicon (Si) for which Z = 14. Explain your answer. Are all the shells filled?
-
Anne always signed up for the most challenging biology courses during her college years. She has a great deal of confidence in her ability to meet the challenges of these courses. Anne is...
-
Write the ground state electron configuration for Nb and Nb3 + .
-
Among the 3 dimensioning techniques A, B and C, which one is correct. 50 50 A B 50 C
-
The standard operating capacity of Tecate Manufacturing Co. is 1,000 units. A detailed study of the manufacturing data relating to the standard production cost of one product revealed the following:...
-
Fabulous Motor Company makes electric cars and has two products, the Simplegreen and the Fabulousgreen. To produce the Simplegreen, Fabulous Motor employed assets of $ 24,500,000 at the beginning of...
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
Saints, Inc., has the following data available for two of its divisions for last year: The tax rate for Saints, Inc., is 30 percent. a. Compute the following for each division: (1) Sales margin (2)...
-
Solve problems involving parallelograms, trapezoids and kites. Write the correct answer in the answer sheet. 1. A cross section of a water trough is in the shape of a trapezoid with bases measuring...
-
Which subshell has the lowest energy? (a) 4s (b) 3p (c) 2p (d) 3s (e) 2s
-
Write the ground-state electron configuration for each of the following atoms or ions. Which have a valence-shell octet? (a) Ar (b) Na + (c) C 2 (d) O 2 (e) Ca 2 +
-
What is the fixed-overhead budget variance?
-
The HNH Corporation will pay a constant dividend of $4 per share, per year, in perpetuity. Assume all investors pay a 25% tax on dividends and that there is no capital gains tax. The cost of capital...
-
EJH Company has a market capitalization of $1.4 billion and 25 million shares outstanding. It plans to distribute $120 million through an open market repurchase. Assuming perfect capital markets: a....
-
FCF Co. has 16,000 shares outstanding and a total market value of $1 million, $350 thousand of which is debt and the other $650 thousand is equity. It is planning a 10% stock dividend. a. What is the...
-
RFC Corp. has announced a $2.30 dividend. If RFCs last price while trading cumdividend is $58, what should its first ex-dividend price be (assuming perfect capital markets)?
-
AMS Company has unexpectedly generated a one-time extra $5 million in cash-flow this year. After announcing the extra cash flow, AMS stock price was $60 per share (it has 1 million shares...
-
What are the three financial-performance measures for investment centers?
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
The fundamental vibrational frequencies for 1 H 2 and 2 D 2 are 4401 and 3115 cm 1 , respectively, and De for both molecules is 7.667 10 19 J. Using this information, calculate the bond energy of...
-
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
The rotational constant for 7 Li 19 F determined from microwave spectroscopy is 1.342583 cm 1 . The atomic masses of 7 Li and 19 F are 7.00160041 and 18.9984032 amu, respectively. Calculate the bond...
-
Henry Mintzberg is a management education guru best known for his categorization of managerial roles. Watch Mintzberg on Managing to learn more about the evolution of management education from...
-
Let V be the solid region inside both the sphere x+12+2 = 1 and the first octant x, y, z 0. When expressed in spherical coordinates (with 02, 0 , and p 20 as usual), V is defined by inequalities...
-
What is the equivalent expression to the following code using array bracket notation: *(x+10) = 'g'?"

Study smarter with the SolutionInn App


