Which of the following are electron-transfer reactions? (a) 2 CrO2 + 2 HCrO72 + HO (b) Fe
Question:
Which of the following are electron-transfer reactions?
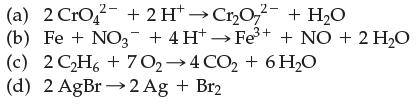
Transcribed Image Text:
(a) 2 CrO2 + 2 H→Cr₂O72 + H₂O (b) Fe + NO3+ 4H+→Fe³+ + NO + 2 H₂O (c) (d) 2AgBr→2Ag + Br2 2C2H6+7O,→4CO,+6H,O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Of the following reactions in the image a 2 CrO22H Cr2O7 HO b Fe NO3 4H Fe3 NO 2 H2O c 2CH7O 4CO 6HO ...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Which of the following proposed reactions would take place quickly under mild conditions? (a) (b) (c) (d) (e) (CHJ2CH-C-NH, + CH3OH (CHJ-CH-C-OCH, + NH, CH,CH,--C-Cl + CH3-C-OH CH,CH-_C_O_C_CH, + HCI
-
The following are electron configurations for some ions. Which ones would you expect to see in chemical compounds? State the concept or rule you used to decide for or against any ion. a. Fe2+...
-
Which of the following reactions occurs most rapidly? Why? a. b. c. Br - H20 C(CH3)3 C(CH3)a (CH)a C(CH3)3 Br - H20 CH33 C(CH3)3
-
Marc & Kent operate a vineyard as partners. The partnership trades under the business name Victor Estate. Victor Estate has prepared a general ledger for the 2023 income year showing the following...
-
What is the first step in preparing a master budget?
-
After researching the different forms of business organization. Natalie Koebel decides to operate Cookie Creations as a proprietorship. She then starts the process of getting the business running. In...
-
In the modified Atwood machine shown in Figure P8.57, each of the three blocks has the same inertia \(m\). One end of the vertical spring, which has spring constant \(k\), is attached to the single...
-
The records of Reuben, Inc., reflect the following data: Work in process, beginning of month2,000 units one-half completed at a cost of $1,250 for materials, $675 for labor, and $950 for overhead....
-
"Imagine you were to open up a fully new coffee shop next to a university, think about the aftermentioned business and answer the following questions: 1. Define the made up firms business model- What...
-
Which of the following are redox reactions? (a) 2Na + 2H 2 O 2NaOH + H 2 (b) MgBr 2 + 2NaF MgF 2 + 2NaBr (c) 2CO + O 2 2CO 2 (d) SO 2 + H 2 O H 2 SO 3
-
Why can we always call an electron-transfer reaction a redox reaction?
-
Consult Sections 204 and 301 of SARBOX. In the post-Sarbanes audit environment, which of the issues that arose in 1996 and 1997 would have to be reported to the audit committee at Sunbeam? Do you...
-
Using this link, https://www.apa.org/workforce/index?_ga=2.244899072.469430581.1660061777-687191231.1660061777 answer the following question... There are only three areas within the field of...
-
1. Ronnie Gilley Properties, LLC (Gilley), wrote a check to Cile way Properties, LLC (Cile), for $100,000. when Cile deposited the check in its bank, it was misencoded in the amount of $1,000. As a...
-
After a series of all-day meetings with software vendors, the CEO, COO and CFO of a hospital evaluated their options and decided on a course of action. As they began implementing the new electronic...
-
A newly married couple is looking for a new 3-bedroom house in London. They surveyed 12 houses they liked and recorded the price and the distance from the nearest station of those houses. The data is...
-
As a rule of thumb, what assets should be used FIRST during retirement so as to minimize the tax liability for spouses/common-law partners? a) non-registered assets of the higher income spouse or...
-
Briefly state Adam Smith's four requirements for a good tax system.
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
A meniscus concave glass (n l = 1.5) thin lens (see Fig. 5.12) has radii of curvature of +20.0 cm and +10.0 cm. If an object is placed 20.0 cm in front of the lens, show that the image distance will...
-
Going back to Section 5.2.3, prove that for a thin lens immersed in a medium of index n m That done, imagine a double-concave air lens surrounded by water; determine if its converging or diverging. (...
-
A biconvex glass (n 1 = 1.5) thin lens is to have a +10.0-cm focal length. If the radius of curvature of each surface is measured to be the same, what must it be? Show that a spider standing 1.0 cm...
-
5. Which of Porter's Five Forces could be most threatening to the long-term success of any business. Explain your answer. (10 marks) 6. Which of the five tasks in the Strategy Making, Strategy...
-
You wish to purchase a $300,000 home. Putting 20% down, you will borrow $240,000 at 6.0%, with monthly payments for 30 years. How much will each payment be? How much interest will be paid over the...
-
There are 300 cars which must travel from city A to city B. There are two possible routes that each car can take. The upper route through city C or the lower route through city D. Let x be the number...

Study smarter with the SolutionInn App


