Which of the following electron transitions in hydrogen would absorb the largest amount of energy? A. B.
Question:
Which of the following electron transitions in hydrogen would absorb the largest amount of energy?
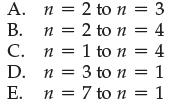
Transcribed Image Text:
A. B. C. D. E. n = 2 to n = 3 n 2 to n = 4 n = 1 to n = 4 n = 3 to n = 1 = n n = 7 to n = 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The electron transition in hydrogen that would absorb the largest amount of energy is E n 1 to n 4 T...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
1. Which of the following electron transitions between two energy states (n) in the hydrogen atom corresponds to the emission of a photon with the longest wavelength? (a) 2 5. (b) 5 2. (c) 5 8....
-
Without doing detailed calculations, indicate which of the following electron transitions requires the greatest amount of energy to be absorbed by a hydrogen atom: from (a) n = 1 to n = 2; (b) n = 2...
-
Without doing detailed calculations, indicate which of the following electron transitions in the hydrogen atom results in the emission of light of the longest wavelength. (a) n = 4 to n = 3; (b) n =...
-
Imagine you are a member of your local school board. You wantan answer to the very simple question, Are students learning?What can you recommend to school principals to answer thequestion?
-
Metropolis Manufacturing Company manufactures a small electric motor that is a replacement part for the more popular gas furnaces. The standard cost card shows the product requirements as follows:...
-
Turfpro Company produces lawn mowers and purchases 4,500 units of a rotor blade part each year at a cost of $ 30 per unit. Turfpro requires a 15% annual rate of return on investment. In addition, the...
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A gene that is normally expressed in pancreatic cells was cloned and then subjected to...
-
For 2010, Sampsell Inc. computed its annual postretirement expense as $240,900. Sampsells contribution to the plan during 2010 was $180,000. Prepare Sampsells 2010 entry to record postretirement...
-
What is the relationship between voltage drop and the resistance in the circuit? Explain.
-
Consider a 1s 2s electron transition. For which atom would this require shorter wavelength light, H or He? Justify your answer.
-
Predict the formula for the compound aluminum nitride made from the elements aluminum and nitrogen, and explain how you made your prediction.
-
Palomar Paper Products purchased land in 2002 for $15,000 cash. The company has held the land since that time. In 2020, Palomar purchased another tract of land for $ 15,000 cash. Assume that prices...
-
Write an equation that includes the building blocks of the nominal rate of interest.
-
Assume the real risk-free rate of interest is 3%, while inflation is expected to be 3% for the next 2 years. If a 2-year Treasury note yields 6.5%, what is the maturity-risk premium for this 2-year...
-
Nadja from Austria wants to make a single investment and have $100,000 after her graduation in 5 years. She did found a microfinance organization that offered her 15 percent per annum; however, Nadja...
-
Maria wants to buy an apartment in the city center after her graduation in 4 years. An apartment costs $20,000 and the price is expected to increase by 4 percent each year. Maria wants to sell her...
-
In September 1963, the first issue of the comic book X-MEN was issued. The original price for that issue was $0.12. By September 2016, 53 years later, the value of the near-mint copy of this comic...
-
When transfer prices are based on actual cost, a supplying division often has no incentive to reduce cost. For example, a design change that would reduce the supplying division's manufacturing cost...
-
Shreemaya Hotel in !adore was facing a problem of low demand for its rooms due to off season. The Managing Director (MD) of the hotel, Mrs. Sakina was very worried. She called upon the Marketing...
-
The muzzle velocity of a rifle bullet is 890.m s 1 along the direction of motion. If the bullet weighs 35 g, and the uncertainty in its momentum is 0.20%, how accurately can the position of the...
-
Why is the probability of finding the harmonic oscillator at its maximum extension or compression larger than that for finding it at its rest position?
-
Why does the energy of a rotating molecule depend on l , but not on m l ?
-
Angel D. Company has net working capital of S350,000 that it expects to grow at a rate of 10% per year forever. Mark N., CFO of the company, is considering some suggestions that could slow that...
-
Jacob's Jewelers is considering carrying a new product line which is expected to produce annual sales of $450,000 and increase cash expenses by $305,001. If the product line is added, taxes will...
-
A quadratic equation can have either one or two distinct real or complex roots depending upon nature of discriminant of the equation. Where discriminant of the quadratic equation is given by A =...

Study smarter with the SolutionInn App


