Would you expect these two molecules to have nearly identical properties? H 2, H
Question:
Would you expect these two molecules to have nearly identical properties?
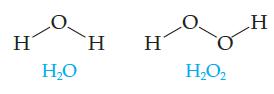
Transcribed Image Text:
Η Η Ο Ἡ H Η2Ο, H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The molecules H 2 O water and H 2 O 2 hydrogen peroxide h...View the full answer

Answered By

Irfan Ali
I have a first class Accounting and Finance degree from a top university in the World. With 5+ years experience which spans mainly from the not for profit sector, I also have vast experience in preparing a full set of accounts for start-ups and small and medium-sized businesses. My name is Irfan Ali and I am seeking a wide range of opportunities ranging from bookkeeping, tax planning, business analysis, Content Writing, Statistic, Research Writing, financial accounting, and reporting.
4.70+
249+ Reviews
530+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
You have two samples of water, each made up of different isotopes of hydrogen: one contains and the other, a. Would you expect these two water samples to be chemically similar? b. Would you expect...
-
You have two identical containers, one containing gas A and the other gas B. The masses of these molecules arc m A = 3.34 X 10-27 kg and m B = 5.34 X 10-26 kg.Both gases arc under the same pressure...
-
Cyclopropane (C3H6) has the shape of a triangle in which a C atom is bonded to two H atoms and two other C atoms at each corner. Cubane (C8H8) has the shape of a cube in which a C atom is bonded to...
-
96. A 66-year-old woman with a long history of heavy smoking presents to her doctor with complaints of shortness of breath and chronic coughing that has been present for about 2 years and has been...
-
If you became the new manager at a restaurant with high employee turnover, what actions would you take to increase retention of employees?
-
Mattvilles general fund had several interfund activities during the fiscal year ended June 30, 20X9 (see the left-hand column of the following table). The right-hand column lists the types of...
-
The Howell Corporation has the following account balances (in millions): Prepare an income statement and a supporting schedule of cost of goods manufactured for the year ended December 31, 2017. (For...
-
The debits to Work in ProcessRoasting Department for Morning Brew Coffee Company for August 2014, together with information concerning production, are as follows: All direct materials are placed in...
-
Mercedes-Benz AG is a subsidiary of Daimler AG known for producing luxury vehicles and commercial vehicles. This company has strongly pushed for product placement in Hollywood movies. In 2015,...
-
Work together to finish the following statement: You dont need Greek numerical prefixes when naming simple ionic compounds because . . .
-
Predict the formula of the compound that forms between phosphorus (P) and hydrogen.
-
Explain several reasons for termination of leases.
-
Antoinettes coastal property in Mississippi has five major ponds that she uses to raise shrimp. The productivity of each pond follows: Assume that each pond is the same size and that the variable...
-
XYZ plc is considering purchasing new equipment that would cost 40 million. The expected unlevered cash flows from this investment are 13 million per annum for the next 5 years. The companys stock...
-
The shareholders of Flannery SA have voted in favour of a buyout offer from Stultz Corporation. Information about each firm is given here: Flannerys shareholders will receive one share of Stultz...
-
Read the paper by M. Massari, F. Roncaglio and L. Zanetti, On the Equivalence between the APV and the WACC Approach in a Growing Leveraged Firm, European Financial Management, Vol. 14, No. 1, 2008,...
-
The matrix in Figure 1 shows payoffs based on the strategies chosen by two firms. If they collude and hold prices at $10, each firm will earn prof its of $5 million. If A cheats on the agreement,...
-
Partial productivity measures should be calculated only for high-value-added activities. Do you agree?
-
Assume a simple Keynesian depression economy with a multiplier of 4 and an initial equilibrium income of $3,000. Saving and investment equal $400, and assume full employment income is $4,000. a. What...
-
a. What is the average time required for H 2 to travel 1.00 meter at 298 K and 1 atm? b. How much longer does it take N 2 to travel 1 m on average relative to H 2 under these same conditions? c....
-
As mentioned in Section 33.3, the only differences between the quantities mp , ave , and rms involve constants. a. Derive the expressions for ave and v rms relative to mp provided in the text....
-
At what temperature is the rms of Ar equal to that of SF 6 at 298 K? Perform the same calculation for mp .
-
The IRR of the following cash flows is Year 0 1 0723 Cash Flow -$10,104 5,500 4,700 4.900
-
If you were to complete an balanced scorecard and wanted to share it with staff what methods do you think would be most effective to educating staff?
-
Assignment2A: Finding multiples We've learned that in programming, there is a special operator for getting the whole number remainder of division: The Modulo Operator! So, 11 % 10 will give us a...

Study smarter with the SolutionInn App


