You have seen that the 4s subshell fills before the 3d subshell. This being true, what would
Question:
You have seen that the 4s subshell fills before the 3d subshell. This being true, what would be wrong with drawing the Bohr diagram as follows?
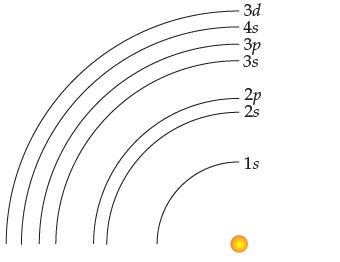
Transcribed Image Text:
NO 1s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The image youve shared appears to be a representation of the electron energy levels in an atom often referred to as electron shells or orbitals In the ...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
You have applied for a job with a local bank. As part of its evaluation process, you must take an examination on time value of money analysis covering the following questions: a. Draw time lines for...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
You have seen that the larger the sample size, the smaller the sampling error tends to be in estimating a population mean by a sample mean. This fact is reflected mathematically by the formula for...
-
Consider the following molecule: Tin II Chloride(SnCl2) Are the bonds polar or non-polar? Is the molecule polar or non-polar?
-
Roberts Corporation uses a process cost system. The records for the month of May show the following information: Required: Prepare a cost of production summary for eachdepartment. Production Report...
-
Elm Corporation and Maple Company have announced terms of an exchange agreement under which Elm will issue 8,000 shares of its $10 par value common stock to acquire all of Maple Companys assets. Elm...
-
In 2014, Javier Ramos decided to open seafood restaurants in California and Texas. Ramos claims that he was describing the crusted glaze applied to cooked seafood to his friend Ivan Murillo when...
-
Clarke Inc. operates the Patio Furniture Division as a profit center. Operating data for this division for the year ended December 31, 2014, are as shown below. In addition, Clarke incurs $180,000 of...
-
You recently joined a prestigious venture capital firm in Munich. As one of your first tasks, you should analyze the performance of the following mutually exclusive investment opportunities by...
-
Bohr solved the potassium problem by putting its last electron where? How did he justify this?
-
How many electrons can each subshell hold before it is considered full?
-
What are the mechanisms through which wealthy individuals and large corporations influence the political system?
-
Assume that the risk-free rate is 3 per cent and the expected return on the FTSE 100 index is 9 per cent. The standard deviation of the market index is 23 per cent. You are managing the pension fund...
-
The Dutch firm, ABS Equipment, has an investment opportunity in the United Kingdom. The project costs 12 million and is expected to produce cash flows of 2.7 million in year 1, 3.5 million in year 2,...
-
You are the finance director of a British company which is expecting a payment in euros of 200 million at the end of September and wish to hedge against currency risk. However, the nearest maturity...
-
You plan to raise funds through following Islamic principles. You require funding today of 3 billion Bahrain dinars and would like to pay it back in equal amounts over 10 years in monthly...
-
Suntharee Lhaopadchan is a Thai student who is planning a one-year stay in the United Kingdom. She expects to arrive in the United Kingdom in 8 months. She is worried about depreciation in the Thai...
-
What are some important behavioral and implementation issues in strategic performance measurement? How does the management accountant deal with these issues?
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
In explaining molecular structure, the MO model uses the change in MO energy with bond angle. Explain why the decrease in energy of the 1 a 1 and 2 a 1 MOs as 2 decreases more than offsets the...
-
The angular functions, Π(θ)Φ(), for the one-electron HartreeFock orbitals are the same as for the hydrogen atom, and the radial functions and radial probability...
-
What is the in-plane amplitude of the wave functions describing the Ï network in the conjugated molecules shown in Figures 24.18 and 24.19? a-2B a-B a+B a+B a+2B a-2B a+2B- m=3 m=4 m=5 m=6...
-
analyze the events prior to financial meltdown of 2008. What are the causes of this event? Use a research format template (Introduction, Statement of Problem, Root Cause Effect, Proposed Solution,...
-
A test charge of +1C is placed halfway between a charge of +3.1C and another of +6.5 separated by 10 cm. What is the magnitude of the force (in Newtons) on the test charge? Your answer should be a...
-
Three long, straight wires are located at the corners of an equilateral triangle of side L= 20 cm as shown in the figure above. I = 4A, I2 = 12 A, I3 = 5A. a) What is the magnetic field due to I1 and...

Study smarter with the SolutionInn App


