Balance each of the following chemical equations by inspection. (a) Sn(s) + P(s) Sn3P(s) (b) Fe(CO3)3(s)
Question:
Balance each of the following chemical equations by inspection.
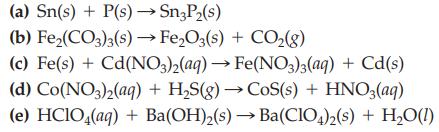
Transcribed Image Text:
(a) Sn(s) + P(s) →→→Sn3P₂(s) (b) Fe₂(CO3)3(s) → Fe₂O3(s) + CO₂(g) (c) Fe(s) + Cd(NO3)2(aq) → Fe(NO3)3(aq) + Cd(s) (d) Co(NO3)2(aq) + H₂S(g) → CoS(s) + HNO3(aq) (e) HClO4(aq) + Ba(OH)₂(s)→ Ba(ClO4)2(s) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Equation a Sns Ps Sn3P2s There is one tin Sn atom on the left and three on the rightso multiply Sn o...View the full answer

Answered By

Poonam Chaudhary
I have 15 month+ Teaching Experience
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Balance each of the following chemical equations by inspection. (a) PCl 5 (s) + H 2 O(l) H 3 PO 4 (aq) + HCl(aq) (b) TiCl 4 (s) + H 2 O(g) TiO 2 (s) + HCl(g).
-
Balance each of the following chemical equations by inspection. (a) F 2 (g) + NaBr(aq) Br 2 (l) + NaF(aq) (b) Sb 2 S 3 (s) + HCl(aq) SbCl 3 (aq) + H 2 S(g).
-
Balance each of the following chemical equations by inspection. (a) FeO(l) + Al(l) Al 2 O 3 (l) + Fe(l) (b) MnO 2 (l) + Al(l) Al 2 O 3 (l) + Mn(l).
-
Aoslia is a small country that takes the world price of corn as given. Its domestic supply and demand for corn are given by the following: a. Assume initially that Aoslia does not open to trade. What...
-
Mindy Kaliny Company has the following internal control procedures over cash disbursements. Identify the internal control principle that is applicable to each procedure. 1. Company checks are...
-
What is deep learning? How is it different from machine learning? How do these relate to AI?
-
LiveJournal was a popular social media platform which enabled online communities to post comments and content related to particular themes. Its most popular community was called Oh No They Didnt!...
-
Gloria Bush has performed a sampling plan to estimate the number of children per household in her neighborhood. In doing so, she established a 10 percent acceptable level of sampling risk and found a...
-
4. The atomic numbers of elements, U, V, W and X are 13, 14, 16 and 17 respectively. Which one of the elements will react with oxygen to form an ionic compound? (Atomic number of oxygen is 8) X A. B....
-
Classify each reaction in Exercise 17 as one of the following: combination, decomposition, single replacement, double replacement, or neutralization. Exercise 17 Balance each of the following...
-
Balance each of the following chemical equations by inspection (a) Pb(s) + O(g) PbO(s) (b) LINO3(s)LiNO(s) + O(g) (c) Mg(s) + HCHO(aq) Mg(CHO)2(aq) + H(g) (d) Hg2(NO3)2(aq) + NaBr(aq) HgBr(s) +...
-
The adjusted trial balance of Rachel Rey Dance Company at April 30, 2016, follows: Requirements 1. Prepare Rachel Reys multi-step income statement for the year ended April 30, 2016. 2. Journalize...
-
function by where Let (X, d) b a compact metric space and a > 0. We define the following M av m || || Ca (X;R) R VC (XR) ||f||a = ||f|| + sup {\f(z) - 5(!)); 2,4 X, x + 1}. f(y)] x, y 0 [d(x, y)]*...
-
Weatherwear estimates that every unit sold and returned due to defect costs the company $200 in profits. Approximately what would Weatherwears total profits have been in Year 5 if all units sold and...
-
While the monetarists say that the source of the change in nominal GDP is the money supply, Keynesians suggest that this increase in nominal GDP depends on the change in public expenditure. According...
-
To expand the storage capacity of highly enriched uranium stockpiles, the Company has won a contract to store canisters containing U-235 in the General Materials Storage Warehouse. Your task is to...
-
The average annual wage (in thousands of dollars) for teachers in a country at various grade levels is shown in matrix W. A small school district has both city schools and county schools; matrix N...
-
Refer to Exhibit 12.6 in the text. What is the depreciation expense deduction in each of four years for a $10,000 asset classified under MACRS as three-yearproperty? Class 3-year property 5-year...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Depletion ComputationsTimber Jonas Lumber Company owns a 7,000-acre tract of timber purchased in 2003 at a cost of $1,300 per acre. At the time of purchase the land was estimated to have a value of...
-
Depletion ComputationsMining Henrik Mining Company purchased land on February 1, 2010, at a cost of $1,250,000. It estimated that a total of 60,000 tons of mineral was available for mining. After it...
-
Depletion ComputationsMinerals At the beginning of 2010, Callaway Company acquired a mine for $850,000. Of this amount, $100,000 was ascribed to the land value and the remaining portion to the...
-
You have been hired as a data analyst for the company. Adventure Works managers have requested that you analyze 3 years of sales and cost data to help them answer specific business questions. In the...
-
How would a data analyst evaluate a conference call to get a feel for the sentiment of management?
-
Need help with this relational model, please. I am having trouble understanding how to create the relational model.

Study smarter with the SolutionInn App


