Balance each of the following chemical equations by inspection (a) Pb(s) + O(g) PbO(s) (b) LINO3(s)LiNO(s)
Question:
Balance each of the following chemical equations by inspection
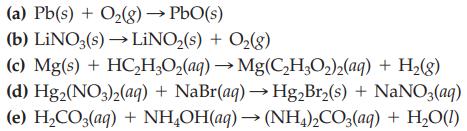
Transcribed Image Text:
(a) Pb(s) + O₂(g) → PbO(s) (b) LINO3(s)→→LiNO₂(s) + O₂(g) (c) Mg(s) + HC₂H₂O₂(aq) → Mg(C₂H₂O₂)2(aq) + H₂(g) (d) Hg2(NO3)2(aq) + NaBr(aq) → Hg₂Br₂(s) + NaNO3(aq) (e) H₂CO3(aq) + NH₂OH(aq) → (NH4)2CO3(aq) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Pbs O2g PbOs Count the number of atoms of each element on both sides of the equationThere is 1 lead Pb atom on the left and 1 lead Pb atom on the ri...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Balance each of the following chemical equations by inspection. (a) PCl 5 (s) + H 2 O(l) H 3 PO 4 (aq) + HCl(aq) (b) TiCl 4 (s) + H 2 O(g) TiO 2 (s) + HCl(g).
-
Balance each of the following chemical equations by inspection. (a) F 2 (g) + NaBr(aq) Br 2 (l) + NaF(aq) (b) Sb 2 S 3 (s) + HCl(aq) SbCl 3 (aq) + H 2 S(g).
-
Balance each of the following chemical equations by inspection. (a) FeO(l) + Al(l) Al 2 O 3 (l) + Fe(l) (b) MnO 2 (l) + Al(l) Al 2 O 3 (l) + Mn(l).
-
Compute the indicated quantities for the given homomorphism. Ker () for : S 3 Z 2 in Example 13.3 Data from Example 13.3 Let S n be the symmetric group on n letters, and let : S n Z 2 be defined by...
-
While examining cash receipts information, the accounting department determined the following information: opening cash balance $150, cash on hand $1,125.74, and cash sales per register tape $990.83....
-
In connection with deep learning, what is backpropagation?
-
The Church of God at Southaven entered into an agreement with a general contractor called National Church Services (NCS) for the construction of a sanctuary for about \($1.1\) million. NCS hired...
-
Axis Systems specializes in servers for work- group, e-commerce, and enterprise resource planning (ERP) applications. The company's original job cost system has two direct cost categories: direct...
-
7. The full symbol of an ion of an element is 39X+? Which one of the following is the number of electrons in the ion? A. 18 B. 19 C. 20 D. 39 8. The atomic numbers of elements W, X, Y and Z are 9,...
-
Balance each of the following chemical equations by inspection. (a) Sn(s) + P(s) Sn3P(s) (b) Fe(CO3)3(s) FeO3(s) + CO(g) (c) Fe(s) + Cd(NO3)2(aq) Fe(NO3)3(aq) + Cd(s) (d) Co(NO3)2(aq) + HS(g) ...
-
Balance each of the following chemical equations by inspection. (a) Co(s) + O(g) CoO3(s) (b) LiClO3(s) LiCl(s) + O(g) (c) Cu(s) + AgCHO(aq) Cu(CHO)2(aq) + Ag(s) (d) Pb(NO3)2(aq) + LiCl(aq) PbCl(s)...
-
In 2014, TLI Communications, LLC, filed a lawsuit against a multitude of companies such as Apple, DropBox, Facebook, Google, Instagram, and Yahoo!. The lawsuit alleged the defendants had violated...
-
State the domain of the given function. 6 f(x)= 6x-4
-
Happyville's population can be modeled using the function P (t) = 31000e0.17, where P (t) is the population t years after January 1st, 2007. In what year is the population of Happyville expected to...
-
To be successful and understand if Four Seasons is meeting its goals, you need to develop a marketing plan overview that is specific about what Marketing Goals and Objectives are along with the...
-
Consider SC(X), the safe configuration space for two robots on the space X shown on the right in Figure 6.36. (a) Sketch a representation of SC(X), showing how it can be constructed by gluing...
-
Calculate for Each Project: NPV (at 12%) IRR MIRR (at 12%) Pl (at 12%) Profitability Index = NPY/(absolute value of Initial Investment) Crossover point calculation: Project Rankings: NPV Rankings IRR...
-
Given the following information, calculate the amount of after-tax profit for the period: sales, $260; expenses other than depreciation, $140; depreciation, $50; marginal income tax rate, 35 percent....
-
What can scientists learn by comparing the fossilized skeletons of extinct primates with the bones of modern species?
-
Ratio Analysis the 2007 Annual Report of Eastman Kodak contains the following information. Compute the following ratios for Eastman Kodak for 2007. (a) Asset turnover ratio. (b) Rate of return on...
-
Book vs. Tax (MACRS Depreciation) Annunzio Enterprises purchased a delivery truck on January 1, 2010, at a cost of $41,000. The truck has a useful life of 7 years with an estimated salvage value of...
-
Book vs. Tax (MACRS Depreciation) Elwood Inc. purchased computer equipment on March 1, 2010, for $36,000. The computer equipment has a useful life of 10 years and a salvage value of $3,000. For tax...
-
How to create a relational model from DDL statements and how you save a relational model design.?
-
Please convert this E-R diagram to Relational model. Production Fixed Expenses FixedExpenseID DatePaid Description Amount Category Item Show/Production ShowID Actors Budget SetBudget OpiningDate...
-
Provide three comparisons between hierarchical, network and relational database models. You can present your answer in the table. (b) The relational database model is the most widely used database....

Study smarter with the SolutionInn App


