Calculate the atomic mass for iron given the following data for its natural isotopes. 54Fe: 56Fe: 57
Question:
Calculate the atomic mass for iron given the following data for its natural isotopes.
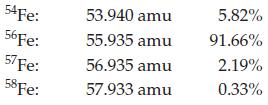
Transcribed Image Text:
54Fe: 56Fe: 57 Fe: 58 Fe: 53.940 amu 55.935 amu 56.935 amu 57.933 amu 5.82% 91.66% 2.19% 0.33%
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
To calculate the atomic mass of iron Fe youll use the weighted aver...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Modify the code to add another attribute and a method that fits (is consistent with) the object the class is modeling, or suggest a different version of the code. Write the main method to demonstrate...
-
Calculate the atomic mass for zinc given the following data for its natural isotopes. 64Zn 66Zn 67Zn 68 Zn 70Zn 63.929 amu 65.926 amu 66.927 amu 67.925 amu 69.925 amu 48.89% 27.81% 4.11% 18.57% 0.62%
-
Calculate the atomic mass for magnesium given the following data for its natural isotopes. 24 Mg: 25Mg: 26Mg: 23.985 amu 24.986 amu 25.983 amu 78.70% 10.13% 11.17%
-
In Exercises 1126, determine whether each equation defines y as a function of x. x + y = 25
-
Explain the limitations of using operating leverage to predict profitability.
-
Suppose you are considering renting an apartment. You, the renter, can be viewed as an agent while the company that owns the apartment can be viewed as the principal. What agency conflicts do you...
-
The cells that carry oxygen to body tissues are (a) red blood cells. (b) white blood cells. (c) platelets. (d) hemoglobin.
-
Laurie Gladin owns land and a building that she has been using in her sole proprietorship. She is going to incorporate her sole proprietorship as a C corporation. Laurie must decide whether to...
-
10. The graph was created by placing a motion detector below a 100.0g mass oscillating up and down while hanging on a spring of unknown spring constant, 10 -10 m N 0 0.6 1.2 1.8 2.4 3.0 time (s) a)...
-
Which has the longer wavelength: red light or yellow light?
-
Calculate the atomic mass for lithium given the following data for its natural isotopes. 'Li: 7Li: 6.015 amu 7.016 amu 7.42% 92.58%
-
All diamonds are made of carbon. Write the negation of the statement.
-
Why is it important to understand (and assess) internal controls?
-
What does the auditor do when they identify control exceptions?
-
Name a generally accepted framework used to describe internal controls.
-
Name the four different techniques for testing controls.
-
Why is it important to identify both the strengths and weaknesses in a system of internal controls?
-
Samara Brothers, Inc. (Samara), is a designer and manufacturer of children's clothing. The core of Samara's business is its annual new line of spring and summer children's garments. Samara sold its...
-
Consider the combustion of methanol below. If 64 grams of methanol reacts with 160 grams of oxygen, what is the CHANGE in volume at STP. 2CH3OH(g) + 3O2(g) 2CO2(g) + 4H2O(1) The volume decreases by...
-
What are the direct and indirect costs of an IPO? MINI CASE Randys, a family-owned restaurant chain operating in Alabama, has grown to the point where expansion throughout the entire southeast is...
-
What are equity carve-outs? MINI CASE Randys, a family-owned restaurant chain operating in Alabama, has grown to the point where expansion throughout the entire southeast is feasible. The proposed...
-
In what other ways are investment banks involved in issuing securities? MINI CASE Randys, a family-owned restaurant chain operating in Alabama, has grown to the point where expansion throughout the...
-
Match the event to the expected exchange rate effect between Japan (whose currency is the yen) and South Africa (whose currency is the rand). Assume each has a floating exchange rate policy. 1....
-
Suppose France imports more from Britain than Britain imports from France (because the French really like English bread and cheese!) What type of Balance of Payments imbalance is this?
-
One of the two major airlines serving a city has decreased the price of its fights. What impact wil his have on demand for car rentals in the city? Willingness to pay for car rentals will increase by...

Study smarter with the SolutionInn App


