Classify each reaction in Exercise 19 as one of the following: combination, decomposition, single replacement, double replacement,
Question:
Classify each reaction in Exercise 19 as one of the following: combination, decomposition, single replacement, double replacement, or neutralization.
Exercise 19
Balance each of the following chemical equations by inspection
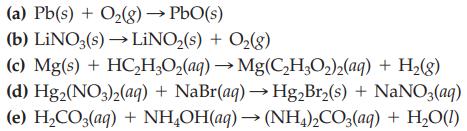
Transcribed Image Text:
(a) Pb(s) + O₂(g) → PbO(s) (b) LINO3(s)→→LiNO₂(s) + O₂(g) (c) Mg(s) + HC₂H₂O₂(aq) → Mg(C₂H₂O₂)2(aq) + H₂(g) (d) Hg2(NO3)2(aq) + NaBr(aq) → Hg₂Br₂(s) + NaNO3(aq) (e) H₂CO3(aq) + NH₂OH(aq) → (NH4)2CO3(aq) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Pbs O2g PbOs Combination reaction This is because two elements lead and oxygen com...View the full answer

Answered By

Rishabh Ojha
During my undergraduate i used to participate as TA (Teaching Assistant) in several electronics and computers subject. I'm passionate about learning Computer Science as my bachelors are in Electronics but i learnt most of the Computer Science subjects on my own which Machine Learning also. At Present, i'm a working professional pursuing my career as a Machine Learning Engineer and i want to help others learn during my free hours, that's all the motivation behind giving tuition. To be frank i have no prior experience of tutoring but i have solved problems on opensource platforms like StackOverflow and github. ~Thanks
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The IPRC-DFRD-funded pilot project is not going as well as planned. The project manager is quite unsure about the outcome of any present phases. Team members seem to be frustrated about the project...
-
Classify each reaction in Exercise 20 as one of the following: combination, decomposition, single replacement, double replacement, or neutralization. Exercise 20 Balance each of the following...
-
Determine whether the given set of matrices under the specified operation, matrix addition or multiplication, is a group. Recall that a diagonal matrix is a square matrix whose only nonzero entries...
-
At July 31, Martinez Company has the following bank information: cash balance per bank $7,420, outstanding checks $762, deposits in transit $1,120, and a bank service charge $20. Determine the...
-
Go to tobiipro.com/fields-of-use/marketing-consumer-research/. What services and insights can they provide to advertisers?
-
The plaintiff, Dennis Rubel, was permanently injured while working at Lowes Home Center Inc., and hired Michael Dzienny to represent him in a personal injury lawsuit. When discussing a settlement...
-
Identify the journal in which each of the following transactions is recorded. 1. Cash sales 2. Owner withdrawal of cash 3. Cash purchase of land 4. Credit sales 5. Purchase of merchandise on account...
-
18. The atomic number of elements P, Q, R and T are 19,17, 14 and 6 respectively. The pair of elements that can react to form an ionic compound is A. Q and T B. R and Q C. Q and P D. R and T 19. The...
-
Write a balanced equation for each of the following combination reactions. (a) Nickel metal is heated with oxygen gas to produce solid nickel(II) oxide. (b) Iron wool is heated with oxygen gas to...
-
Classify each reaction in Exercise 18 as one of the following: combination, decomposition, single replacement, double replacement, or neutralization. Exercise 18 Balance each of the following...
-
From a survey of 250 coworkers you find that 155 would like the company to provide on-site day care. a) What is the value of the sample proportion p? b) What is the standard error of the sample...
-
Suppose that the demand function for q units of a product priced at Sp per unit is given by P 400(q+1) (q+2) Find the rate of change of price with respect to the number of units.
-
Sunland Inc. issued $150,000, 5-year bond on January 1, 2024 paying 6% interest on a semi-annual basis every January 1 and July 1. Prepare the first-year journal entries for the bond issue and...
-
To what extent do cultural norms influence how other societies and cultures deal with the issue of sexual harassment in the workplace? What are the pros and cons of mediation for an employer? What...
-
Consider an economy with identical households who are distributed on the unit interval. Social planner faces the following problem: subject to max Btu(ct) t=0 (1) Ct +kt+1 = [(1 Xt)kt] + (1 8)kt...
-
A chemist needs 1 0 0 milliliters of a 4 4 % solution but has only 1 6 % and 5 6 % solutions available. Find how many milliliters of each that should be mixed to get the desired solution.
-
Given an asset with a net book value (NBV) of $25,000, what are the after-tax proceeds for a firm in the 34 percent tax bracket if this asset is sold for $35,000 cash? What are the after-tax proceeds...
-
What are the six activities involved in the physical supply/distribution system?
-
Depreciation ComputationAddition, Change in Estimate In 1983, Abraham Company completed the construction of a building at a cost of $1,900,000 and first occupied it in January 1984. It was estimated...
-
DepreciationReplacement, Change in Estimate Peloton Company constructed a building at a cost of $2,400,000 and occupied it beginning in January 1991. It was estimated at that time that its life would...
-
Error Analysis and Depreciation, SL and SYD Kawasaki Company shows the following entries in its Equipment account for 2011. All amounts are based on historical cost. (a) Prepare any correcting...
-
1. a) Suppose you deposit $2500 each year for 20 years in a savings account paying 5% compounded annually. How much would the account contain after 20 years? How much of the FV did you actually...
-
2. Home Depot (HD) recently paid a annual dividend of $1.50 per share. Using a one period dividend growth model, what is the price of one share of HD stock if the terminal dividend growth rate is 5%?...
-
Max Labs Inc. current portfolio of products has a 10% IRR and a 10% standard deviation. The correlation coefficients with the firm portfolio are 0.5 (r=0.5) for both, Dog Treats and Dog Food; zero...

Study smarter with the SolutionInn App


