Classify each reaction in Exercise 20 as one of the following: combination, decomposition, single replacement, double replacement,
Question:
Classify each reaction in Exercise 20 as one of the following: combination, decomposition, single replacement, double replacement, or neutralization.
Exercise 20
Balance each of the following chemical equations by inspection.
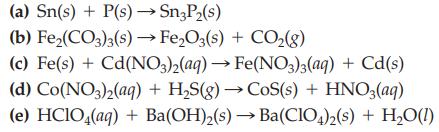
Transcribed Image Text:
(a) Sn(s) + P(s) →→→Sn3P₂(s) (b) Fe₂(CO3)3(s) → Fe₂O3(s) + CO₂(g) (c) Fe(s) + Cd(NO3)2(aq) → Fe(NO3)3(aq) + Cd(s) (d) Co(NO3)2(aq) + H₂S(g) → CoS(s) + HNO3(aq) (e) HClO4(aq) + Ba(OH)₂(s)→ Ba(ClO4)2(s) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Lets classify each reaction based on the type of reaction a Sns Ps Sn3P2s Type ...View the full answer

Answered By

Collins Omondi
I have been an academic and content writer for at least 6 years, working on different academic fields including accounting, political science, technology, law, and nursing in addition to those earlier listed under my education background.
I have a Bachelor’s degree in Commerce (Accounting option), and vast knowledge in various academic fields Finance, Economics, Marketing, Management, Social Science, Women and Gender, Business law, and Statistics among others.
4.80+
4+ Reviews
16+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The IPRC-DFRD-funded pilot project is not going as well as planned. The project manager is quite unsure about the outcome of any present phases. Team members seem to be frustrated about the project...
-
Classify each reaction in Exercise 19 as one of the following: combination, decomposition, single replacement, double replacement, or neutralization. Exercise 19 Balance each of the following...
-
The number of males per 100 females, age 70 or over, in the United States for some recent years is shown in the following table. (a) Plot the data, letting t be the years since 1970. (b) Would a...
-
At August 31, Oscar Company has a cash balance per books of $8,500 and the following additional data from the bank statement: charge for printing Oscar Company checks $35, interest earned on checking...
-
Go to analytics.google.com. What tools are available for a business to analyze its data? Whats involved in the free set-up?
-
A customer sued a pharmacist and the drug store that employed the pharmacist (Walgreens). The pharmacist reviewed the prescription records of a customer (during her shift at work), learned that the...
-
Saturated steam at a gauge pressure of 2.0bar is to be used to heat a stream of ethane. The ethane enters a heat exchanger at 16?C and 1.5 bar gauge at a rate of 795m 3 /min and is heated at constant...
-
The atomic number of element M, N, R, and T are 1, 8, 12 and 14 respectively. The element which can form ions by either losing or gaining electron(s) is M 11. A. B. N C R D. T 12. The atomic number...
-
Write a balanced equation for each of the following combination reactions. (a) Nickel metal is heated with oxygen gas to produce solid nickel(II) oxide. (b) Iron wool is heated with oxygen gas to...
-
Classify each reaction in Exercise 18 as one of the following: combination, decomposition, single replacement, double replacement, or neutralization. Exercise 18 Balance each of the following...
-
What are the trends in this product area and the industry? Discuss the competition.
-
To what extent do socio-cultural factors influence the efficacy of organizational development interventions, and how can practitioners navigate cultural nuances to facilitate cross-cultural synergy...
-
1. You have taken over the management of a paper production firm. You find that the firm is contracted to deliver 3,000, 6,000, 5,000, and 2,000 reams of paper over the next four reams of paper in...
-
Direct materials variances The following data is provided for Bellingham Company: DATA Resources' requirements, standard pol 2.5 Resources' standard price, per pound $3.30 Units produced 15,000...
-
Figure shows a single degree of freedom mechanism which has a motor attached at point M to supply its motion. M Run Rr Lunk 3 8-120 R Link 2 (Slider) Use of a high quality lubricant on the pin at...
-
23. Consider the following 0/1 knapsack problem: Use the bottom-up DP approach to fill the DP table. Find the maximum value that can be put in the knapsack and the optimal solution set (items that...
-
Given the following attributes of an investment project with a five-year life, and an after-tax discount rate of 12 percent, calculate the net present value (NPV) and the payback period of the...
-
Modify the counter from Exercise 5.44 such that the counter will either increment by 4 or load a new 32-bit value, D, on each clock edge, depending on a control signal Load. When Load = 1, the...
-
Composite Depreciation Presented below is information related to Morrow Manufacturing Corporation. (a) Compute the rate of Depreciation per year to be applied to the plant assets under the composite...
-
Depreciation Computations, SYD Pippen Company purchased a piece of equipment at the beginning of 2007. The equipment cost $502,000. It has an estimated service life of 8 years and an expected salvage...
-
DepreciationChange in Estimate Machinery purchased for $52,000 by Carver Co. in 2006 was originally estimated to have a life of 8 years with a salvage value of $4,000 at the end of that time....
-
Keeley and Roy are concerned about their financial future and meeting their goals. They are 38 years old with twin girls, Abigail and Laryssa age 8. Keeley is a public relations account executive...
-
The interest rate is fixed at 10%. You are offered an asset that pays $1,000 from now until eternity at yearly intervals. You find its present value by calculating the sum of the discounted annual...
-
The kibitzer computes the average of these three probabilities and announces the result of his computation publicly. Beatrice and Carol update their probabilities for F in the light of this new...

Study smarter with the SolutionInn App


