Draw and illustrate the voltaic cell described in the practice exercise. Refer to Figure 17.4 and label
Question:
Draw and illustrate the voltaic cell described in the practice exercise. Refer to Figure 17.4 and label the anode and cathode; show the direction of electron and anion flow.
Figure 17.4
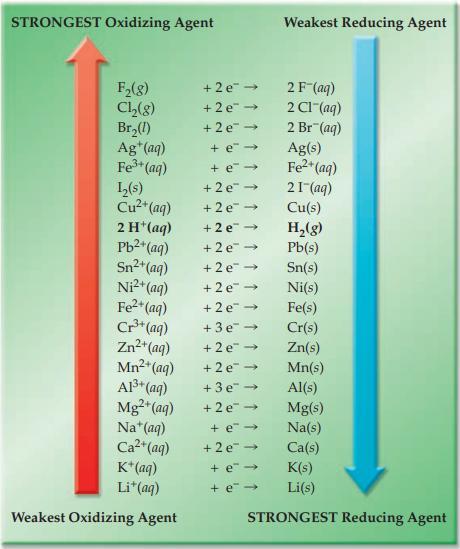
Transcribed Image Text:
STRONGEST Oxidizing Agent F₂(g) Cl₂(g) Br₂(1) Ag+ (aq) Fe³+ (aq) 1₂(s) Cu²+ (aq) 2 H+ (aq) Pb²+ (aq) Sn²+ (aq) Ni²+ (aq) Fe²+ (aq) Cr³+ (aq) Zn²+ (aq) Mn²+ (aq) Al³+ (aq) Mg2+ (aq) Na+ (aq) Ca²+ (aq) K+ (aq) Li* (aq) Weakest Oxidizing Agent + 2 e 2F (aq) +2e →>> 2 C1-(aq) +2e → 2 Br (aq) +e→ + e→ +2e → +2e → +2e → +2e → +2e → +2e → +2e → +3e → +2e → +2e → +3e → +2e → Weakest Reducing Agent +e→ +2e → + e→ + e→ Ag(s) Fe²+ (aq) 21 (aq) Cu(s) H₂(g) Pb(s) Sn(s) Ni(s) Fe(s) Cr(s) Zn(s) Mn(s) Al(s) Mg(s) Na(s) Ca(s) K(s) Li(s) STRONGEST Reducing Agent
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Fes Sn 2 aq Sns Fe 2 ...View the full answer

Answered By

Irfan Ali
I have a first class Accounting and Finance degree from a top university in the World. With 5+ years experience which spans mainly from the not for profit sector, I also have vast experience in preparing a full set of accounts for start-ups and small and medium-sized businesses. My name is Irfan Ali and I am seeking a wide range of opportunities ranging from bookkeeping, tax planning, business analysis, Content Writing, Statistic, Research Writing, financial accounting, and reporting.
4.70+
249+ Reviews
530+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Draw and illustrate the electrolytic cell described in the practice exercise. Refer to Figure 17.6 and label the anode and cathode; show the direction of electron flow. Figure 17.6 Zn anode Zn+ Zn is...
-
Voltaic cell is constructed from the following halfcells: a magnesium electrode in magnesium sulfate solution and a nickel electrode in nickel sulfate solution. The halfreactions are Mg(s) Mg2 +...
-
A voltaic cell is created using silver (Ag / Ag + ) as one electrode and manganese (Mn / Mn 2+ ) as the other electrode. Reduction half-reaction E o (V) Ag + ( aq ) + e - Ag ( s ) 0.80 Mn 2+ ( aq )...
-
Examine how effective change communication can reduce resistance in organization ? explain
-
Should portfolio effects impact the way investors think about the risk of individual stocks?
-
Assume that sin = 0.5812 and cos = 0.2341 and that both and are first-quadrant angles. Evaluate cos ( + ).
-
Craxton Engineering will either purchase or lease a new $756,000 fabricator. If purchased, the fabricator will be depreciated on a straight-line basis over seven years. Craxton can lease the...
-
The Speedy Pizza Palace is revamping its order-processing and pizza-making procedures. In order to deliver fresh pizza fast, six elements must be completed. a. Construct a precedence diagram and...
-
6. The major components in "Gun Metal" are: (a) Cu, Ni and Fe (b) Al, Cu, Mg and Mn (c) Cu, Sn and Zn 7. 8. Which of the following ore is concentrated using group 1 cyanide salt (a) Calamine (b)...
-
Nickel can react with aqueous silver nitrate solution according to the following ionic equation: Ni(s) + 2 Ag + (aq) 2 Ag(s) + Ni 2+ (aq) Assume the half-reactions are separated into two...
-
Predict whether the following reaction is spontaneous or nonspontaneous. Ni 2+ (aq) + Al(s) Ni(s) + Al 3+ (aq)
-
Find the equations of two parabolas that pass through the points (2, 5), (0, 9), and (- 6, 7). Sketch each parabola.
-
True Or False Medical professionals may use waivers to limit their liability.
-
True Or False What constitutes reasonable care in an emergency depends on the circumstances in which the emergency occurs.
-
True Or False Structured settlements minimize the income tax a plaintiff has to pay.
-
Under a comparative-negligence defense, a plaintiff who was awarded $1,000,000 and who was found to be 30 percent negligent would receive an award of ____________.
-
True Or False When a pure comparative-negligence standard is used, the plaintiff can recover no matter how extensive their own negligence.
-
Creed Napier, the regional manager of E-Taxes, Inc., is evaluating the performance of four ecommerce tax preparation sites in her region. The following data for the past six months were presented to...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
Superior Micro Products uses the weighted-average method in its process costing system. Data for the Assembly Department for May appear below: Required: 1. Compute the cost per equivalent unit for...
-
Data concerning a recent periods activity in the Prep Department, the first processing department in a company that uses process costing, appear below: A total of 20,100 units were completed and...
-
Maria Am Corporation uses a process costing system. The Baking Department is one of the processing departments in its strudel manufacturing facility. In June in the Baking Department, the cost of...
-
Briefly discuss how Azure migration tools can facilitate ABC Bank total migration to cloud solution, and what are the steps that the ABC Bank should undertake after the migration of their system to...
-
Supply Chain Management You are responsible to evaluate three bids to supply surge protector outlets for 140 server racks. PowerPort submits a bid of $170 per unit with a defect rate of 3%; SureFlow...
-
Lori Greiner, billionaire Shark and "Queen of QVC " , exclaimed, " Oh my God!," after learning six pieces of vegan chicken wings, costing $ 2 . 6 7 to produce, are sold for $ 2 3 . Based on this...

Study smarter with the SolutionInn App


