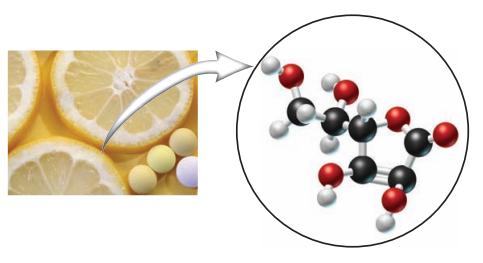
Examine the model for vitamin C and determine the molecular formula. In the model shown, the black
Question:
Examine the model for vitamin C and determine the molecular formula. In the model shown, the black sphere = C atom, white = H atom, and red = O atom.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The molecu...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Solve for the amount of firm ABCs cash given the following data: ABC Current Ratio is equal to 2.5 ABC Quick Ratio is equal to 1.2 ABC Current Liabilities is equal to $12K ABC Credit Sales is equal...
-
Based on what you learned in Chapter 2 about atoms, what part of the atom do you think the spheres in the molecular space-filling models shown in Table 3.1 represent? If you were to superimpose a...
-
Examine the model for aspirin and determine the molecular formula. In the model shown, the black sphere = C atom, white = H atom, and red = O atom.
-
1. The following are selected accounts taken from the adjusted trial balances of the Purell Merchandise company on December 31, 2021: Inventory, January 1, 2021 Selling expenses Loss on sale of...
-
The adjusted trial balance of Cavamanlis Co. as of December 31, 2012, contains the following. Instructions (a) Prepare an income statement.(b) Prepare a statement of retained earnings.(c) Prepare a...
-
List three differences between a corporation and a sole proprietorship or a partnership.
-
Repeat the calculations of Example 6.3, but for \(80 \mathrm{~mol} \%\) of the liquid distilled. Data From Example 6.3:- Suppose the liquid of Example 6.1 [50 mol% n-heptane (A), 50 mol% n-octane...
-
The Porsche Shop, founded in 1985 by Dale Jensen, specializes in the restoration of vintage Porsche automobiles. One of Jensens regular customers asked him to prepare an estimate for the restoration...
-
assumption 1 assume there will be 4000 visits, each visit will yield 1400 of revenue
-
Alum, Al 2 (SO 4 ) 3 , is used in styptic pencils to stop minor bleeding. What is the number of atoms in the chemical formula of alum? (a) 10 (b) 12 (c) 14 (d) 17 (e) 21.
-
Write the chemical formula for vitamin B 6 , which is composed of 8 carbon atoms, 11 hydrogen atoms, 1 nitrogen atom, and 3 oxygen atoms.
-
Review the Section 12.2.3 and find the myFirstPos and myLastPos for each processor for the following the cases. A. when numThreads \(=4\) and numBlocks \(=1500\) B. when numThreads \(=6\) and...
-
How does each of the 3 aspects of TIMWOOD below lead to inefficiency for KOI? How can each be improved to enhance efficiency for KOI? Defects Motion Overprocessing Companies often focus on...
-
Human Resources Planning serves as a preliminary step to all other functions of the HRM. Briefly, explain the key steps involved in HRP.
-
Describe any key sales activities that you will use to build and maintain your sales pipeline. Explain the essential elements of the sales process (i.e., staffing, commissions, and quotas). Company...
-
Create a well-written job description for Human resources assistants the organization can use when hiring employees. Describe the essential functions, qualifications, and other characteristics of the...
-
The planning phase of safety management system (SMS) development is well in progress, and it is now time to develop some goals for your SMS. Based upon your workplace (or your research on a...
-
Cooper Huntington is the manager of the upstate store of a large retailer of farm products. His company is a stable, consistently profitable member of the farming industry. The upstate store is doing...
-
Assessing simultaneous changes in CVP relationships Braun Corporation sells hammocks; variable costs are $75 each, and the hammocks are sold for $125 each. Braun incurs $240,000 of fixed operating...
-
An automobile manufacturer is buying some special tools for $100,000. The tools are being depreciated' by double declining balance depreciation using a 4- year depreciable life and a $6250 salvage...
-
This is the continuation of Problem 12-21. Instead of paying $100,000 cash for the tools, the corporation will pay $20,000 now and borrow the remaining $80,000. The depreciation schedule will remain...
-
A project will require the investment of $108,000 in equipment (sum-of-years' -digits depreciation with a depreciable life of 8 years and zero salvage value ) and $25,000 in raw materials (not...
-
12 H2 + 12 Cl2 Na + H2O Na + 12 CI2 HCI K. = 1.00x105 NaOH + 1/2 H2 K. = 1.00x10-2 Naci K = 1.00x107 = Calculate K For NaOH + HCl-NaCl + H2O = Fe2O3(S) HBr H2O OH 2 Fe3+ + 3 02- Ksp 3 = 1.13x10-86 H+...
-
Grant borrowed $5,300 to remodel his bathroom, he agreed to make 30 monthly payments of $235. He payed it off in 24 months. What is the amount neede to repay the loan in full using the rule of 78?
-
Jared invests $3200 at 3.4%/a compounded quarterly. How long will it take for his investment to grow to $8,000

Study smarter with the SolutionInn App


