Given the only naturally occurring isotope of aluminum is Al-27, determine its mass from the periodic table.
Question:
Given the only naturally occurring isotope of aluminum is Al-27, determine its mass from the periodic table.
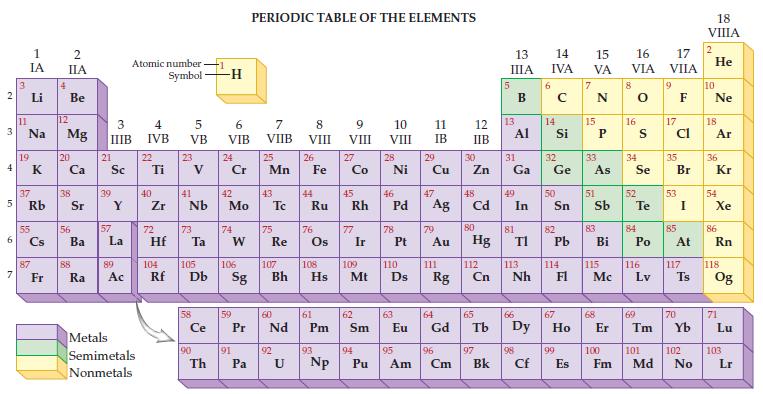
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Isotope Mass 26982amu A...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Given the only naturally occurring isotope of phosphorus is P-31, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB...
-
Given the only naturally occurring isotope of fluorine is F-19, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
Given the only naturally occurring isotope of sodium is Na-23, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
In Exercises 130, find the domain of each function. f(x) = 1 4 x - 2 3
-
Magnificent Modems, Inc., makes modem cards that are used in notebook computers. The company completed the following transactions during 2010. All purchases and sales were made with cash. 1. Acquired...
-
In July 2007, Apple had cash of $7.12 billion, current assets of $18.75 billion, and current liabilities of $6.99 billion. It also had inventories of $0.25 billion. a. What was Apples current ratio?...
-
An article at Mobilize.org reported that the median credit-card balance for undergraduate students was \(\$ 1770\) for those who carried a balance from month to month. A professor at a community...
-
Lasting impressions Co. records all cash receipts on the basis of its cash register tapes. Lasting Impressions Co. discovered during April 2012 that one of its sales clerks had stolen an undetermined...
-
1. If a ball with a mass of 2M moving east at a velocity v collides with a ball with a mass of M moving to the west with a velocity v, which direction will the balls be moving after they collide...
-
Calculate the atomic mass for lithium given the following data for its natural isotopes. 'Li: 7Li: 6.015 amu 7.016 amu 7.42% 92.58%
-
Draw a diagram of the arrangement of protons, neutrons, and electrons in an atom of each of the following isotopes. (a) 31 15 P (b) 35 17 Cl (c) 40 18 Ar (d) 131 5 3 I.
-
The gravitational potential energy of a pendulum is U = mgy? (a) Taking y = 0 at the lowest point, show that y = L (1 cos (), where ( is the angle the string makes with the vertical. (b) If ( is...
-
Why might financial statement users demand an audit?
-
Karl has decided to retain a management reserve on a $400,000 project that includes a $60,000 profit. At the completion of the project, Karl finds that the management reserve fund contains $40,000....
-
Who are the main users of company financial statements?
-
What are four factors that affect inherent risk?
-
What level of detail does the auditor need to include in the audit working papers when documenting the results of their controls testing?
-
Why are industry ethical codes generally ineffective? Explain.
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
What is the net advantage to leasing (NAL)? Does your analysis indicate that Lewis should buy or lease the equipment? Explain. MINI CASE Lewis Securities Inc. has decided to acquire a new market data...
-
Now assume that the equipments residual value could be as low as $0 or as high as $400,000, but that $200,000 is the expected value. Since the residual value is riskier than the other cash flows in...
-
The lessee compares the cost of owning the equipment with the cost of leasing it. Now put yourself in the lessors shoes. In a few sentences, how should you analyze the decision to write or not write...
-
Consider the bar graph of "Media Campaign Coverage by Topic" below. What conclusion can be drawn from the graph? Media coverage of campaigns is a good way for Americans to learn the details of...
-
Refer to the Article Summary. Colorado taxes marijuana with a 2.9% standard sales tax and a 15% state marijuana sales tax, in addition to local taxes. All of these taxes are paid by consumers at the...
-
How does consuming public broadcasting affect one's political knowledge? 1 Group of answer choices Americans who primarily rely on public broadcasting for their news tend to have lower levels of...

Study smarter with the SolutionInn App


