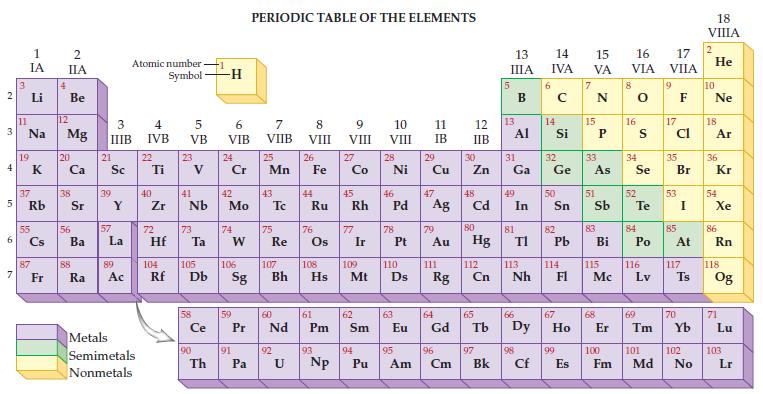
Given the only naturally occurring isotope of sodium is Na-23, determine its mass from the periodic table.
Question:
Given the only naturally occurring isotope of sodium is Na-23, determine its mass from the periodic table.
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Isotope Mass 23 11 Na Abundance 2299 amu 100 Atomic mass of Alu min iu...View the full answer

Answered By

Munir Ahmed Jakhro
I am professional Tutor of of Business Courses, I did my four years Bachelor Degree from one of the Top Business schools of World "Institute of Business Administration" in year 2013. Since then I have been working as Tutor of Accounting, Finance tutor on different online platforms like this website. I am have experience of 6 years teaching business courses to students online and offline my professional job at national savings also helped me in accounting understanding .
4.90+
8+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Given the only naturally occurring isotope of phosphorus is P-31, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB...
-
Given the only naturally occurring isotope of fluorine is F-19, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
Given the only naturally occurring isotope of aluminum is Al-27, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
In Exercises 1318, find the average rate of change of the function from x 1 to x 2 . f(x)=x from x = 4 to x = 9
-
Mantooth Manufacturing Company experienced the following accounting events during its first year of operation. With the exception of the adjusting entries for depreciation, assume that all...
-
JPJ Corp has sales of $1 million, accounts receivable of $50,000, total assets of $5 million (of which $3 million are fixed assets), inventory of $150,000, and cost of goods sold of $600,000. What is...
-
A car requires \(300 \mathrm{~kJ}\) of energy to overcome air resistance and maintain a constant speed of \(20 \mathrm{~m} / \mathrm{s}\) over a distance of \(1.0 \mathrm{~km}\). What is the force of...
-
Sam owns an insurance agency and makes the following business gifts during the year. Calculate Sams deduction for business gifts. Amount Donee Ms. Sears (a client) Mr. Williams (a tennis partner, not...
-
A student uses a scooter to travel around their local area in Brisbane. The scooter and rider together experience a gravitational force of 711 N. When travelling, the system of the scooter-plus-rider...
-
Calculate the atomic mass for lithium given the following data for its natural isotopes. 'Li: 7Li: 6.015 amu 7.016 amu 7.42% 92.58%
-
Draw a diagram of the arrangement of protons, neutrons, and electrons in an atom of each of the following isotopes. (a) 31 15 P (b) 35 17 Cl (c) 40 18 Ar (d) 131 5 3 I.
-
From March 2012 to May 2017, the price of houses increased dramatically in many parts of the country. a. What impact would you expect increases and decreases in home values to have on the consumption...
-
What is an accountability relationship?
-
What is the most appropriate materiality base for a for-profit entity? Why?
-
What might an assurance provider express a conclusion about?
-
What is the purpose of developing an overall audit strategy?
-
What are two examples of assurance providers?
-
What actions are typically forbidden by corporate codes? Explain.
-
Explain the term "Equivalent Units". Why are they calculated in process costing? [4 Marks] [minimum 350 words]
-
Assume that the lease payments were actually $280,000 per year, that Consolidated Leasing is also in the 40 percent tax bracket, and that it also forecasts a $200,000 residual value. Also, to furnish...
-
What do you think the lessors NPV would be if the lease payment were set at $280,000 per year? (Hint: the lessors cash flows would be a mirror image of the lessees cash flows.) MINI CASE Lewis...
-
Lewiss management has been considering moving to a new downtown location, and they are concerned that these plans may come to fruition prior to the expiration of the lease. If the move occurs, Lewis...
-
If you have a job in a "government relations" department, you are a _____________ working for a ______________. Question 61 options: a) government employee; state government b) government employee;...
-
What are interest groups' main goal from policy makers?
-
Suppose some colleagues are having lunch together, and the only White colleague in the group is handed the check at the end of the meal. What does this represent?

Study smarter with the SolutionInn App


