In an automobile collision, sodium azide, NaN 3 , decomposes and fills an air bag with nitrogen
Question:
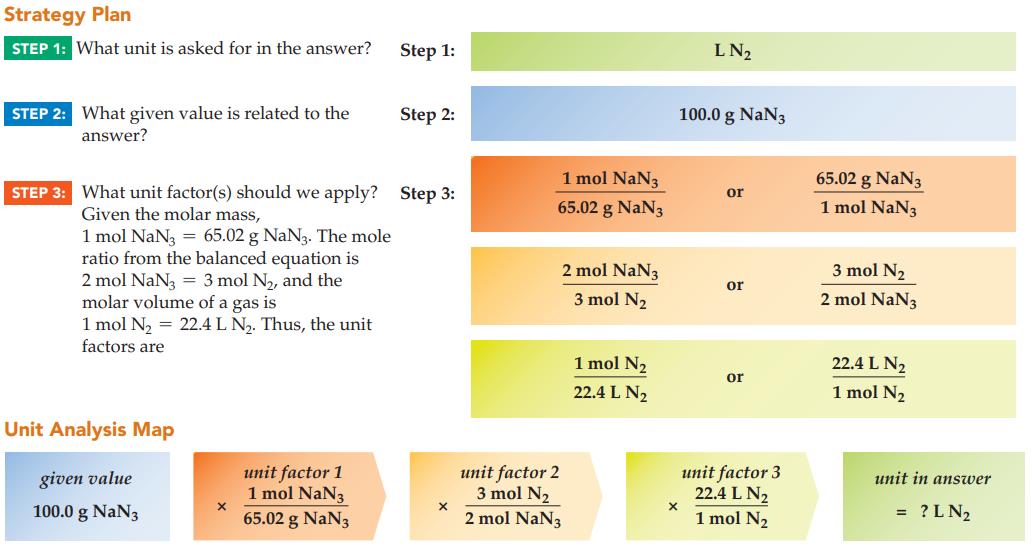
In an automobile collision, sodium azide, NaN3, decomposes and fills an air bag with nitrogen gas. If an air bag contains 100.0 g of NaN3(65.02 g/mol), what is the volume of nitrogen gas produced at STP? The equation for the reaction is
![]()
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:





