Indicate the physical state (solid, liquid, gas) for each of the following at the given temperature. (a)
Question:
Indicate the physical state (solid, liquid, gas) for each of the following at the given temperature.
(a) Ne at -225 °C
(b) Ne at -255 °C
(c) Ar at -175 °C
(d) Ar at -200 °C
The melting points and boiling points for neon and argon are as follows.
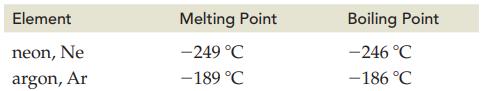
Transcribed Image Text:
Element neon, Ne argon, Ar Melting Point -249 °C -189 °C Boiling Point -246 °C -186 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
ANSWER To determine the physical state of a substance at a given temperature we ...View the full answer

Answered By

Jeff Omollo
As an educator I have had the opportunity to work with students of all ages and backgrounds. Throughout my career, I have developed a teaching style that encourages student engagement and promotes active learning. My education and tutoring skills has enabled me to empower students to become lifelong learners.
5.00+
5+ Reviews
42+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Classify each of the following materials as falling into one of the categories listed in Table 12.2. What particles make up these solids, and what are the forces of attraction between particles? Give...
-
Classify each of the following materials as falling into one of the categories listed in Table 12.2. What particles make up these solids and what are the forces of attraction between particles? Give...
-
Indicate the physical state (solid, liquid, or gas) for each of the following at the given temperature. (a) H 2 O at -10 C (b) H 2 O at 110 C (c) NH 3 at -100C (d) NH 3 at -50 C The melting points...
-
Scott and Associates, Inc., is an accounting firm that has three new clients. Project leaders will be assigned to the three clients. Based on the different backgrounds and experiences of the leaders,...
-
Assuming the same information as for Problem 21-2, suppose Hastings will increase Vandells level of debt at the end of Year 3 to $30.6 million so that the target capital structure is now 45% debt....
-
Two spaceships, each 100 m long when measured at rest, travel toward each other with speeds of 0.85c relative to Earth. (a) How long is each ship as measured by someone on Earth? (b) How fast is each...
-
Wire 1 is \(2.3 \mathrm{~m}\) long and carries a current of \(2.2 \mathrm{~A}\) to the right. Wire 2 is also \(2.3 \mathrm{~m}\) long, and it carries a current of 3. 0 A to the left. The wires are...
-
The accounts receivable department at Rick Wing Manufacturing has been having difficulty getting customers to pay the full amount of their bills. Many customers complain that the bills are not...
-
Kericho Sports Club has been operating for some time now. The treasurer analysed the cash book and produced the following Receipts and Payments account for the year ended 31st Dec. 2018. Receipts...
-
Classify each of the following crystalline solids as ionic, molecular, or metallic: (a) Iodine, I 2 (b) Silver iodide, AgI.
-
Classify each of the following crystalline solids as ionic, molecular, or metallic: (a) Nickel, Ni (b) Nickel(II) oxide, NiO.
-
A spinless particle is scattered by a weak Yukawa potential where > 0 but V 0 can be positive or negative. It was shown in the text that the first-order Born amplitude is given by (a) Using f (1) ()...
-
Explain 4 factors that can influence the supply and demand of loanable funds which are responsible for the variation of the risk-free rates across countries
-
At the end of t years, the future value of an investment of $42,500 in an account that pays 12% compounded monthly is S=42,500 1 + 500 (1+0.12) 12 future value of the investment be more than...
-
Janet buys a one-year put and a one-year call on a share of XYZ stock according to the following information: option put call strike price $55 $55 option premium $4.13 $6.75 XYZ does not pay any...
-
In the C++ programming language, one can do file I/O via so-called streams. In the operation ostream < < 42 < < "universe", ostream is an output stream, and < < is a binary infix operator that...
-
Six years ago, Junk Removal Services (JRS) issued high-yield bonds at par with a maturity of 12 years and a face value of $1,000. Today, the bonds just paid their semi-annual coupons of $70. 20. At...
-
Emelio and Charita are married taxpayers with 2 dependent children. Emelio starts a computer consulting business in 2011. Charita works as a real estate broker. During 2011, they have the following...
-
Describe a job you have had in the past or a job you are very familiar with. Indicate the negative aspects of the job and how it could be improved with current human resource management techniques.
-
Figure shows that r D increases as the debt?equity ratio increases. In MM?s world r E also increases but at a declining rate. Explain why. Redraw Figure, showing how r D and change for increasingly...
-
Imagine a firm that is expected to produce a level stream of operating profits. As leverage is increased, what happens to (a) The ratio of the market value of the equity to income after interest? (b)...
-
Archimedes Levers is financed by a mixture of debt and equity. You have the following information about its cost of capital: Can you fill in the blanks? 12% 'D = 12% TE = BE = 1.5 10% BA D/V = .5 Bo...
-
Talal Properties need a construction machinery costing AED 45,000. The machine will work for 5 years. Expected net cash inflows [in AED] are y1: 30,000; y2: 25,000; y3 :15,000; y4: 10,000 and y5:...
-
1) Describe any two of the following terms concepts: Expectancy theory of motivation. Maslow's need hierarchy. Recognition need. 2)Define any two of the following terms : Autocratic...
-
write a solution of the initial- 4. 3. boundary value problem. 2. 1. uku for 0 < x < L,t> 0. = u(0,1) = u(L,t) = 0, u(x, 0) = x(L-x) u =4ux for 0 < x < L,t> 0, u(0,1) = u(L,t) = 0, u(x, 0) = x(L-x) u...

Study smarter with the SolutionInn App


