Refer to a periodic table and write the predicted electron configuration for each of the following elements.
Question:
Refer to a periodic table and write the predicted electron configuration for each of the following elements.
(a) Zn
(b) Se.
Periodic Table:
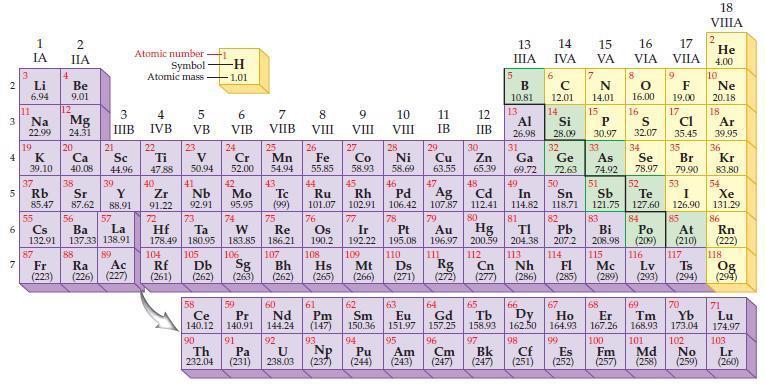
Transcribed Image Text:
2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 R Rb 4 87 2 IIA Be 9.01 12 Mg 24.31 K Ca Sc 39.10 40.08 44.96 20 38 21 3 IIIB 39 Sr Y 85.47 87.62 88.91 57 55 56 La Cs Ba 132.91 137.33 138.91 89 88 Fr Ac Ra (223) (226) (227) Atomic number Symbol Atomic mass 4 IVB 22 Ti 47.88 40 Zr 2 91.22 72 5 VB 104 23 V 50.94 41 73 Hf Ta 178.49 180.95 105 Rf Db (261) (262) -H -1.01 6 VIB Nb Mo 92.91 95.95 90 24 Cr 52.00 42 74 106 Sg (263) 58 Pr Ce 140.12 140.91 91 59 7 VIIB Th Pa 232.04 (231) 25 W Re 183.85 186.21 Mn 54.94 43. Tc (99) 75 107 Bh (262) 60 Nd 144.24 92 U 238.03 8 VIII 26 Fe 55.85 76 Os 190.2 61 Pm (147) 93 Np 9 VIII (237) 10 VIII 11 IB 12 IIB 27 28 29 Zn Co Ni Cu 58.93 58.69 63.55 65.39 44 49 45 46 48 Ru Rh Pd Cd In 101.07 102.91 106.42 107.87 112.41 114.82 Ag 77 78 79 80 Ir Pt 192.22 195.08 109 110 Hs Mt Ds Rg Cn (265) (266) (271) (272) (277) Au Hg 196.97 200.59 111 112 108 47 30 5 13 ΠΙΑ B 10.81 13 6 14 IVA с 12.01 14 15 16 17 VA VIA VILA 7 N 14.01 15 66 67 62 63 64 65 Sm Eu Gd Tb Dy Ho 150.36 151.97 157.25 158.93 162.50 164.93 99 94 95 96 97 Pu Am Cm Bk (244) (243) (247) (247) 8 O 16.00 16 Al Si P 30.97 31 34 26.98 28.09 32 33 Ga Ge As Se 69.72 72.63 74.92 78.97 50 51 52 53 Sn Sb Te I 118.71 121.75 127.60 126.90 82 83 84 85 TI Pb Bi Po At 204.38 207.2 208.98 (209) (210) 113 114 115 116 117 Nh Fl Lv Ts (286) (285) (289) (293) (294) 81 Mc 9 F 19.00 17 CI 35.45 S 32.07 35 Br 79.90 68 69 70 Er Tm Yb 167.26 168.93 173.04 100 101 102 Cf Es Fm Md No (251) (252) (257) 98 (258) (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39,95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 1s 2 2s 2 2p 6 3s 2 3...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to a periodic table and write the predicted electron configuration for each of the following elements by counting the number of electrons in each block: (a) P (b) Co. Periodic Table: 2 3 4 5 6...
-
Refer to the periodic table and write the predicted electron configuration for each of the following elements using core notation. (a) W (b) Bi (c) Ra (d) Ac. Periodic Table: 2 3 4 10 6 3 7 11 Li...
-
Refer to the periodic table and write the predicted electron configuration for each of the following elements using core notation. (a) Sr (b) Ru (c) Sb (d) Cs. Periodic Table: 2 3 4 10 6 3 7 11 Li...
-
What are the concepts of traditional and contemporary organizational design? Will these designs be influenced differently by management and the environment?
-
What are the required financial statements for? (a) A not-for-profit health care entity and (b) A governmental health care entity reporting only business-type activities?
-
What are the key features of a bond? Sam Strother and Shawna Tibbs are vice presidents of Mutual of Seattle Insurance Company and codirectors of the companys pension fund management division. An...
-
In a photoelectric effect experiment, the intensity of the light is increased while the frequency, which is above the threshold frequency, is held constant. As a result, A. There are more electrons....
-
While examining cash receipts information, the accounting department determined the following information: opening cash balance $150, cash on hand $1,125.74, and cash sales per register tape $988.62....
-
George invests 2 4 5 0 dollars at a simple interest rate of 1 0 . 2 percent. How much is his investment worth after 1 9 months?
-
Refer to the periodic table and predict the number of 5d electrons in a Pt atom. Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 87 4 Fr (223) 2 IIA Be 9.01 12 Mg...
-
State the energy sublevel being filled in each of the following series of elements: (a) Cs - Ba (b) Y - Cd (c) In - Xe (d) Ce - Lu.
-
Suppose a liberal arts student is interested in exploring graduate school in his or her field. The student identifies a program in which he or she is interested and finds the name of a few students...
-
Find the following F 1;2; values. (a) v = 10, V2 = 10, and = .05 Vi (b) v = 15, v = (c) v = 12, v = V2 (d) v = 20, V2 = 3, and x = .01 15, and x = .025 10, and x = .005
-
A credit manager at the Bargain Basement Department Store is interested in the proportion of customers who pay their credit card balances in full each month. A random sample of 200 customers...
-
This industry dates back thousands of years, is visible in TV news, is global in nature, and has annual sales of $100 billion. Yet participants do not even agree on how to label it, and most...
-
Reconstruct the confidence interval for the mean home prices given in question 18, but this time construct a 99 % confidence interval. What happens to the size of the confidence interval? Question 18...
-
A quality control engineer believes that the life of light bulbs for his company is normally distributed with a standard deviation of 100 h. A random sample of 10 light bulbs gives the following...
-
Examine the Minitab output shown here for a multiple regression analysis. How many predictors were there in this model? Comment on the overall significance of the regression model. Discuss the t...
-
Use integration by parts to evaluate the following. Check your answer by taking the derivative. x2e-xdx
-
What are some of the key obstacles for the FASB and IASB in their convergence project for the statement of cash flows?
-
Wainwright Corporation had the following activities in 2010. 1. Sale of land.............................................................$180,000 2. Purchase of...
-
Stansfield Corporation had the following activities in 2010. 1. Payment of accounts payable.............................$770,000 2. Issuance of common stock..................................$250,000...
-
Define the Definition and Scope of Credit Analysis, Principles Informing Credit Analysis. (Analyzing quantitative measures: important financial ratios, liquidity, leverage, and profitability to build...
-
class UnaryExpression : public Expression { public: UnaryExpression(Expression* operand); double evaluate(); private: Expression* operand; }; class BinaryExpression : public Expression { public:...
-
Discuss the extent to which you believe the academic literature has been successful in designing early warning systems that signal impending crisis situations and outline the difficulties involved in...

Study smarter with the SolutionInn App


