Refer to a periodic table and write the predicted electron configuration for each of the following elements
Question:
Refer to a periodic table and write the predicted electron configuration for each of the following elements by counting the number of electrons in each block:
(a) P
(b) Co.
Periodic Table:
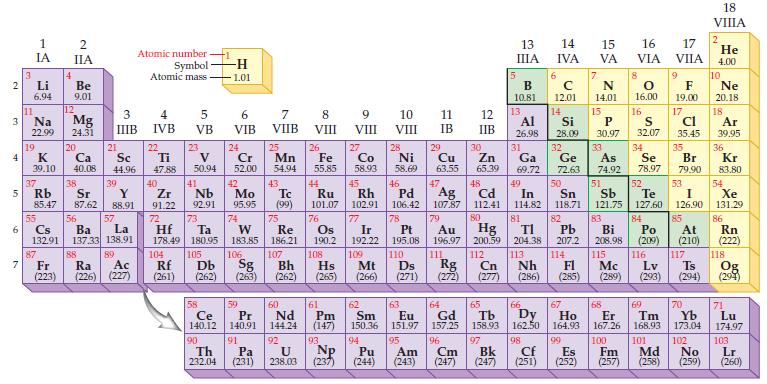
Transcribed Image Text:
2 3 4 الا 5 6 7 3 Li 6.94 11 1 IA Na 22.99 19 37 G 85.47 4 55 2 IIA Be 9.01 38 Rb Sr 87 12 K Ca Sc 39.10 40.08 44.96 Mg 24.31 20 21 87.62 3 IIIB 39 56 Cs Ba 132.91 137.33 138.91 Y 88.91 57 Lo La 89 O 88 Fr Ac Ra (223) (226) (227) Atomic number Symbol Atomic mass 4 IVB 22 Ti 47.88 40 Zr 91.22 72 5 VB 23 V 50.94 41 Nb 92.91 73 Hf Ta 178.49 180.95 104 105 ᎠᏏ Rf (261) (262) 58 Ce 140.12 90 Th 232.04 -H 1.01 6 VIB 24 Cr 52.00 42 7 VIIB 25 Mn 54.94 Tc Mo 95.95 (99) 91 43 74 75 W Re 183.85 186.21 Pa (231) 59 60 Pr Nd 140,91 144.24 8 VIII 92 U 238.03 26 Fe 55.85 44 Ru 101.07 76 77 Ir 192.22 106 107 109 Sg Bh Hs Mt (263) (262) (265) (266) 108 Os 190.2 9 VIII 93 27 Np (237) Co 58.93 10 11 VIII IB 28 Ni 58.69 45 46 Rh Pd 102.91 106.42 29 Cu 63.55 47 12 IIB 30 Zn 65.39 61 62 63 64 65 Pm Sm Eu Gd Tb (147) 150.36 151.97 157.25 158.93 94 95 Pu Am (244) (243) 78 Pt 195.08 110 Ds Rg Cn (271) (272) (277) 111 13 IIIA 96 97 Cm Bk (247) (247) 5 C B 10.81 12.01 13. 48 Ag 107.87 79 Cd 112.41 80 Au Hg Pb 196.97 200.59 204.38 207.2 112 113 Nh FI (286) (285) 114 Al 26.98 14 IVA 6 F 14 15 31 Ga 69.72 49 Si P 28.09 30.97 32 33 34 Ge As Se 72.63 74.92 78.97 50 51 52 In Sn Sb Te 114.82 118.71 121.75 127.60 81 82 83 84 Bi Po 208.98 (209) 115 116 Cf (251) 66 67 Dy Ho 162.50 164.93 98 99 7 15 17 16 VA VIA VIIA N 14.01 8 0 16.00 16 68 Er 167.26 100 Es Fm (252) (257) S 32.07 9 F 19.00 17 cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 117 Mc Lv (289) (293) (294) 18 VIIIA 69 70 Tm Yb 168.93 173.04 101 102 Md No (258) (259) 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 118 Ts Og (294) Rn (222) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Now that you understand blocks of elements in the periodic table you can predict the order of sublev...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to a periodic table and write the predicted electron configuration for each of the following elements. (a) Zn (b) Se. Periodic Table: 2 3 4 15 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 R Rb 4 87 2...
-
Refer to the periodic table and write the predicted electron configuration for each of the following elements using core notation. (a) W (b) Bi (c) Ra (d) Ac. Periodic Table: 2 3 4 10 6 3 7 11 Li...
-
Refer to the periodic table and write the predicted electron configuration for each of the following elements using core notation. (a) Sr (b) Ru (c) Sb (d) Cs. Periodic Table: 2 3 4 10 6 3 7 11 Li...
-
A curve C in three dimensions is given parametrically by (x(t), y(t), z(t)), where t is a real parameter, with a t b. Show that the equation of the tangent line at a point P on this curve where t...
-
Financial StatementsPrivate University. The following is the pre-closing trial balance for Horton University as of June 30, 2011. Additional information related to net assets and the statement of...
-
How do flotation costs affect the cost of capital? Are these costs about the same for each of the there capital components? How do they change as the firm raises larger and larger amounts of capital,...
-
A light sensor is based on a photodiode that requires a minimum photon energy of \(1.7 \mathrm{eV}\) to create mobile electrons. What is the longest wavelength of electromagnetic radiation that the...
-
At Webster Chemical Company, lumps in the caulking compound could cause difficulties in dispensing a smooth bead from the tube. Even when the process is in control, an average of four lumps per tube...
-
First, write the value(s) that make the denominator(s) zero. Then solve the equation. 15 +3=15
-
Which energy sublevel is being filled by the elements Sc to Zn? (a) 3 d (b) 4 s (c) 4 p (d) 4 d (e) 4 f.
-
State the highest energy sublevel in each of the following elements: (a) H (b) S (c) Ni (d) U.
-
What is the expected effective yield of the investment portfolio?
-
The personnel department wants to estimate the cost of hiring a new secretary. The following data are collected on 8 new secretaries: Estimate the average cost of hiring. Construct a 90 % confidence...
-
The manager in the local supermarket wanted to know whether it is worth the trouble to keep the store open 24 h a day. He randomly sampled and recorded 20 nights sales and got Use M1NITAB to answer...
-
In question 87, what is the 95 % confidence interval for the variance of the soda pumped into the cans? Question 87 A soft drink producer installs a new assembly line to fill 12-oz soda cans. After a...
-
In question 85, if the dean wants to narrow the 90 % confidence interval to 1 %, how many students should he ask? Question 85 The dean of student activities wants to estimate the average spending on...
-
What distinguishes intellectual property from other types of property, such as land, buildings, and inventory? Provide several examples of intellectual property and describe their importance to a...
-
Use the following data to determine the equation of the multiple regression model. Comment on the regression coefficients. Predictor Coefficient Constant .......31,409.5 x1 ......... .08425 x2...
-
Describe basic managerial approaches to implementing controls and how these are implemented.
-
Loveless Corporation had the following 2010 income statement. Revenues............................$100,000 Expenses.................................60,000...
-
Hendrickson Corporation reported net income of $50,000 in 2010. Depreciation expense was $17,000. The following working capital accounts changed. Accounts...
-
In 2010, Wild Corporation reported a net loss of $70,000. Wilds only net income adjustments were depreciation expense $81,000, and increase in accounts receivable $8,100. Compute Wilds net cash...
-
It is important for a public health professional to show evidence of professional development over time. A professional portfolio is one way to collect and showcase achievements and artifacts or...
-
A stock is currently selling for $600 per share. The next year's (t = 1) estimated earnings per share (EPS) of the company is $24. The required rate of return on the stock is 10%. The company will...
-
Initial Evaluation of Concentration Risk in a Simulated Portfolio Extends the financial analysis to evaluate concentration risk within a simulated investment portfolio, broadening the perspective....

Study smarter with the SolutionInn App


