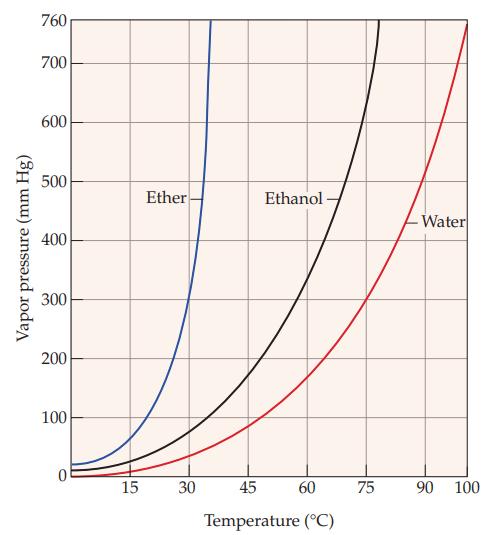
Refer to Figure 11.5 and estimate the vapor pressure of ethanol at each of the following temperatures:
Question:
Refer to Figure 11.5 and estimate the vapor pressure of ethanol at each of the following temperatures:
(a) 45 °C
(b) 60 °C
Figure 11.5
Transcribed Image Text:
Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15 Ether 30 Ethanol- 45 60 Temperature (°C) 75 -Water 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Ethanol at 45C The curve for ethanol intersects the 45C temperature line at approximately 3...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Estimate the vapor pressure of ethanol at temperatures of T = 0, 50, 100, and 150C, using the following methods. A. The Antoine equation B. The Clausius-Clapeyron equation with H vap = 42.0 kJ/mol...
-
Using the information in Problems 7.13 and 8.20, estimate the heat of vaporization for the first bit of ethanol from ethanol-water solutions containing 25, 50, and 75 mol % ethanol and from a...
-
The vapor pressure of ethanol (C2H5OH) at 20C is 44 mmHg, and the vapor pressure of methanol (CH3OH) at the same temperature is 94 mmHg. A mixture of 30.0 g of methanol and 45.0 g of ethanol is...
-
Suppose the weight of pieces of passenger luggage for domestic airline flights follows a normal distribution with u = 28 pounds and o = 5.7 pounds. (a) Calculate the probability that a piece of...
-
Is there a difference between the amount of a parents net income under the equity method and the consolidated net income for the same parent and its subsidiaries?
-
A student wants to work out the answer to the following calculation: 3 + 5 4 He writes the following steps: 3 + 5 4 = 84 =32 a. (i) Explain what mistake he has made. (ii) What should the correct...
-
A \(100-\mathrm{mm}\)-long metal rod is placed in a uniform magnetic field with the rod length perpendicular to the field direction (Figure P29.26). The rod moves at \(0.20 \mathrm{~m} /...
-
A Question of Ethics New Orleans Paddlewheels, Inc. (NOP), is a Louisiana corporation formed in 1982, when James Smith, Sr., and Warren Reuther were its only share holders, with each holding 50...
-
(a) Explain four reasons why multinational corporations (MNCs) forecast exchange rates. (b) (4 marks) SPL Limited, a company based in Kenya expects to receive 2 million Euros in one year's time. The...
-
What is the general relationship between the vapor pressure of a liquid and its temperature?
-
Predict which liquid in each pair has the higher surface tension: (a) CH 3 COOH or C 2 H 5 Cl (b) C 2 H 5 OH or CH 3 OCH 3 .
-
What kinds of functions of local government are usually protected by immunity?
-
The table shows Jill's utility schedule for pancakes and coffee total utility MU from total utility MU from # cups of #pancakes from pancakes last pancake from coffee coffee last cup of coffee L 2 10...
-
Defined benefit obligation, 31 December 20X1 Benefits paid to retirees in 20X2 during the year Current service cost for 20X2 Actual return on plan assets for 20X2 Fair value of plan assets, 31...
-
Active slip plane inclinations are higher than passive slip plane inclinations (True/False) Bishop's simplified method requires assumption of factor of safety and slip surfaces (True/False) Active...
-
You are a staff accountant at a confectionary company, Philly Chocolates International (the Company or PCI). The lease on the current multifunction copiers the Company has in its headquarters is...
-
Yeats, Irons, and Rhoades (1975) found that annual deposit growth for 48 commercial banks can be modeled by the following function: D_(t+1)/D_t=1.172-0.125t^-1+1.135t^-2 where D is the year-end...
-
Determine the correct IRS percentage table, recovery period, and applicable convention for each of the following assets: a. Helicopter b. 68-unit apartment building c. The new Wings Field baseball...
-
What is removed during each of the three stages of wastewater treatment: primary, secondary, and tertiary? During which state would you expect items to be recovered that were accidentally flushed,...
-
In the UK initial public offerings of common stock are usually sold by an offer for sale. Mr. Bean has observed that on average these stocks are underpriced by about 9 percent and for some years has...
-
Get a hold of the prospectus for a recent IPO. How do the issue costs compare with? (a) Those of the Marvin issue and (b) Those shown in Table 15.3? Can you suggest reasons for the differences?...
-
Why are the costs of debt issues less than those of equity issues? List the possible reasons.
-
Company XYZ just paid a dividend of $1 per share. The required return on its stock is 15% (EAR). 1) The annual dividends grow at 10% per year over the next three years. Thereafter, dividends will...
-
15 -x+7 if x < -8 if 8x <5 6. Let f(x) = 3 if x = 5 7 if x > 5 Find the following limits if they exist algebraically.
-
Company ABC plans to issue a 15-year coupon bond. The coupon rate is 3% paid semi-annually and the par value is $1000. To determine the bond price, the company refers to the existing bonds in the...

Study smarter with the SolutionInn App


